
FDA Breakthrough Device Designation Marks Major Milestone for Gene Solutions’ SPOT-MAS 10 Multi-Cancer Screening Test
Key finding:
SINGAPORE, May 27, 2026 /PRNewswire/ — Gene Solutions, a global biotechnology company advancing accessible genomic solutions for cancer detection and precision oncology, today announced that the U.S. Food and Drug Administration has granted Breakthrough Device Designation to SPOT-MAS 10, its multi-omic blood test designed to detect cancer-associated signals.Authors:
GENE SOLUTIONS SGP PTE. LTD.Publisher:PR Newswire
Publication date:28 May 2026
Gene Solutions Showcases AI-Powered Multi-Omics Liquid Biopsy Innovations at TJCC 2026: Advancing Precision Oncology in Taiwan
Key finding:
TAIPEI, May 22, 2026 /PRNewswire/ — Gene Solutions, an innovative global biotechnology company advancing AI-powered multi-omics solutions for cancer detection and precision oncology, participated in the 30th Taiwan Joint Cancer Conference (TJCC 2026), marking its first major oncology conference presence in Taiwan.Authors:
Gene Solutions Genomics Pte LtdPublisher:PR Newswire
Publication date:22 May 2026
Gene Solutions Announces Strategic Entry into Türkiye with Robust Real-World Evidence at the 2nd International Hereditary Cancers Congress 2026
Authors:
Gene Solutions Genomics Pte LtdPublisher:PR Newswire
Publication date:04 March 2026
Comprehensive validation of a cell-free DNA-based assay for multi-cancer detection: A Vietnamese longitudinal prospective cohort study (K-DETEK)
Authors:
Le Son Tran¹˒², Dang Luu-Hong Nguyen¹˒², Hanh Thi-Hue Nguyen¹˒², et al.Publisher:ASCO Breakthrough
Publication date:09 August 2024
triSure NIPT Debuts in Taiwan, Expanding Non-Invasive Prenatal Screening Beyond Aneuploidy
Key finding:
TAIPEI, April 14, 2026 /PRNewswire/ — At the prestigious Taiwan Association of Obstetrics and Gynecology (TAOG) 2026 Annual Meeting—the nation’s largest obstetrics and gynecology society established in 1961— Gene Solutions Taiwan officially introduced triSure, a revolutionary non-invasive prenatal screening technology.Authors:
Gene Solutions Genomics Pte LtdPublisher:PR Newswire
Publication date:14 April 2026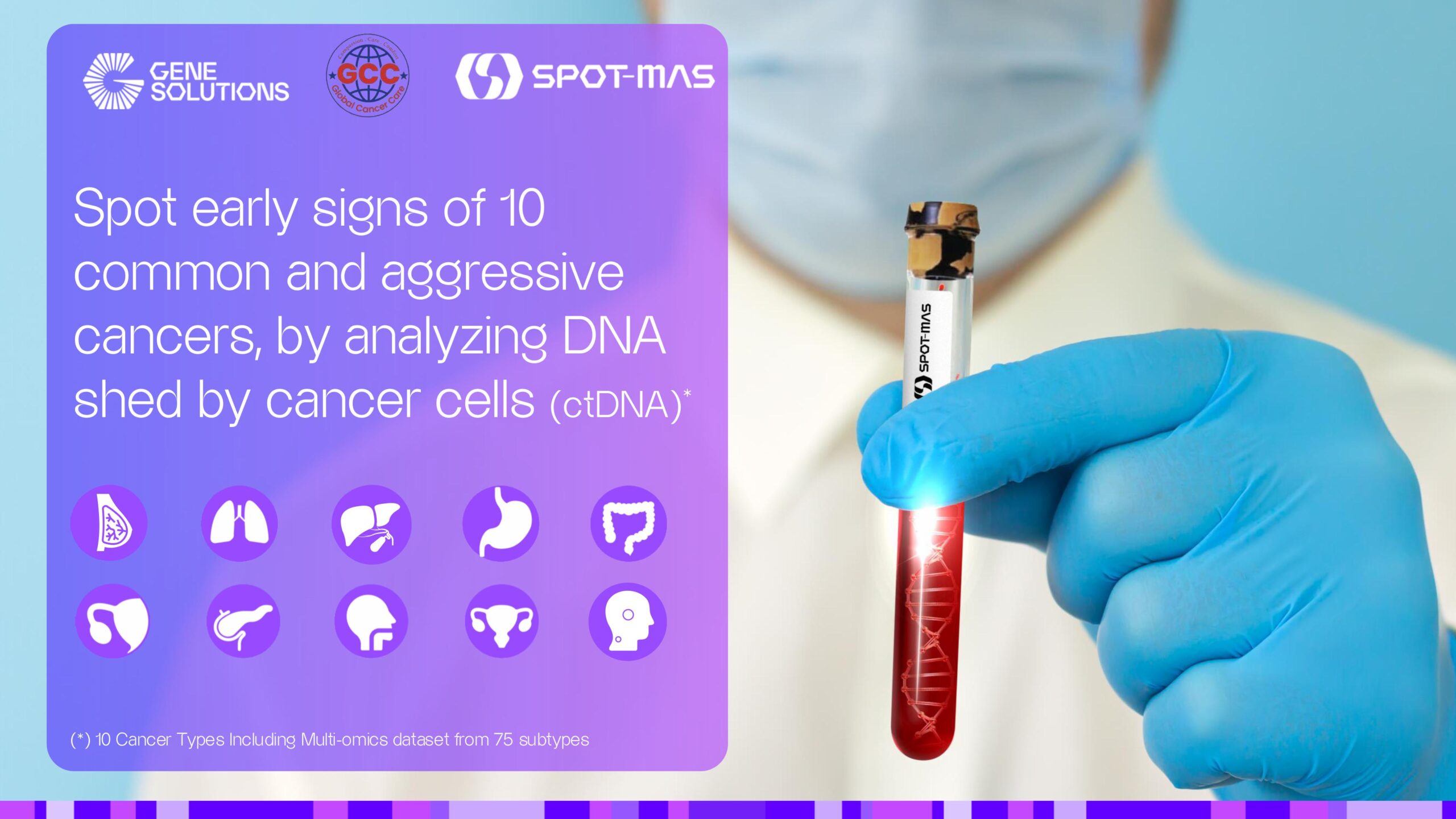
World Cancer Day: Genomics & AI Blood Test Detects Cancer Before Symptoms Appear in India
Publisher:Gene Solutions
Publication date:05 February 2026
Gene Solutions and Pangea Laboratory Establish Strategic Collaboration to Validate Laboratory Developed ctDNA Tests for Precision Screening and Cancer Care in the U.S.
Key finding:
IRVINE, Calif., Jan. 21, 2026 /PRNewswire/ — Gene Solutions, a global biotechnology company, and Pangea Laboratory, a CLIA-certified, CAP-accredited Next Generation Sequencing (NGS) leader in validation platforms for Laboratory Developed Tests (LDTs), announced a strategic collaboration to advance U.S. verification and clinical validation of liquid biopsy assays designed for cancer detection, profiling, and monitoring.Authors:
Gene Solutions Genomics Pte LtdPublisher:PR Newswire
Publication date:21 January 2026
Gene Solutions Concludes Successful ESMO Asia Congress 2025 with AI-Powered Multi-Omics Liquid Biopsy Symposium and Two Best Poster Awards
Key finding:
SINGAPORE, Dec. 20, 2025 /PRNewswire/ — Gene Solutions, a pioneering genetic testing company, announced key achievements at the European Society for Medical Oncology (ESMO) Asia Congress 2025—held in Singapore on December 5–7. Highlights included a full-house symposium, seven scientific abstracts with two earning Best Poster designations, and meaningful engagement with thousands of oncologists from across the Asia-Pacific region at the company’s exhibition booth.Authors:
Gene Solutions Genomics Pte LtdPublisher:PR Newswire
Publication date:20 December 2025
Tumor-naïve multimodal profiling of circulating tumor DNA to detect minimal residual disease in solid tumors
Key finding:
The tumor-naïve method could be a reliable alternative to monitor ctDNA when obtaining high-quality tissue samples is challenging. The performance of this method was better in high ctDNA-shedding cancer or at the metastatic stage.Authors:
Tu Nguyen, Van-Anh Nguyen Hoang, Trong Hieu Nguyen, Trung Hieu Tran, Ngoc Nguyen, Tho Thi Le Vo, Duy Sinh Nguyen, Hoa Giang, Hoai-Nghia Nguyen, and Lan N. TuPublisher:Therapeutic Advances in Medical Oncology
Publication date:18 November 2025
Tumor Genomic and Transcriptomic Analysis Integrated With Liquid Biopsy ctDNA Monitoring: Analytical Validation and Clinical Insights
Key finding:
Comprehensive genomic profiling (CGP) is a time- and tissue- efficient method to help guide precision oncology. To enhance the clinical utility of CGP, we investigated the performance of a novel strategy integrating tumor DNA and mRNA profiling, together with liquid biopsy ctDNA monitoring. Comprehensive genomic and transcriptomic profiling could reliably unveil genetic details not provided by DNA-only CGP. The integration of ctDNA detection further helped detect tumor-agnostic mutations and monitor treatment response.Authors:
Nam H. B. Tran, Thien-Phuc Hoang Nguyen, Vinh Quang Bui, Vu Thuong Le, Trong Khoa Mai, Van-Anh Nguyen Hoang, Tien Anh Nguyen, Minh-Duc Nguyen, Ha-Hieu Pham, Tho Thi Le Vo, My T. T. Ngo, Du Quyen Nguyen, Duy Sinh Nguyen, Hoai-Nghia Nguyen, Minh-Duy Phan, Hoa Giang, Lan N. TuPublisher:Cancer Medicine
Publication date:08 December 2025
SPOT-MAS Launches in Taiwan: AI-Powered Multi-Cancer Early Detection Ushers in a New Era of Precision Medicine
Key finding:
TAIPEI, Nov. 28, 2025 /PRNewswire/ — Precision medicine is entering a new era, driven by breakthroughs in genetic technology and Artificial Intelligence (AI). Multi-Cancer Early Detection (MCED) has become a global trend, and Taiwan joins this movement with the introduction of SPOT-MAS – an advanced screening solution developed by Gene Solutions, a global leader in precision oncology and genomics, in collaboration with local partner Gene Health.Authors:
Gene Solutions Genomics Pte LtdPublisher:PR Newswire
Publication date:28 November 2025
AI and Multi-omics are Transforming Cancer Care: APAC Precision Oncology Conference Outlines the Future of Early Detection and Personalized Treatment
Key finding:
HONG KONG, Nov. 17, 2025 /PRNewswire/ — The APAC Precision Oncology Conference 2025, themed Artificial Intelligence (AI) & Multi-omics in Cancer Management, convened in Hong Kong on November 1, marking Lung Cancer Awareness Month with a clear message: AI and multi-omics are no longer theoretical – they are driving real-world advancements in cancer care across the Asia-Pacific (APAC) region.Authors:
Gene Solutions Genomics Pte LtdPublisher:PR Newswire
Publication date:01 November 20251
2
3
4
5
…
8
