AI and ctDNA Technology: Make Cancer Screening More Accessible and Affordable
Cancer remains a significant health challenge in Asia, accounting for nearly half of the world’s cases. Lung, breast, and colorectal cancers are among the most prevalent, with many patients diagnosed at advanced stages when treatments are less effective and more costly (1). This makes cancer screening not just important—it’s urgent.
While current cancer screening methods have improved survival rates, they have notable limitations. Low sensitivity and specificity can result in missed cancers (false negatives) (2) or unnecessary interventions (false positives) (3), causing patient anxiety (2). Additionally, the invasive nature of some screening methods can deter regular check-ups (4).
The Promise of ctDNA Technology and Multi-Cancer Early Detection (MCED) Tests
Circulating tumor DNA (ctDNA) technology is transforming early cancer detection. ctDNA refers to DNA fragments released by cancerous or pre-cancerous cells into the bloodstream. Since all types of cancer release DNA into the blood, analyzing these fragments through a simple, non-invasive blood draw enables the early detection of multiple cancers, even before clinical symptoms or imaging evidence appears (5).
In 2024, the FDA approved the first blood test for primary colon cancer screening in average-risk individuals, setting a new precedent for blood-based cancer detection (6). This milestone underscores the growing acceptance of ctDNA in cancer screening, for single cancer type or for multiple cancer types.
Numerous Multi-Cancer Early Detection (MCED) clinical studies have been conducted globally, providing robust clinical evidence. In 2018, Gene Solutions developed the SPOT-MAS MCED test, which completed clinical validation through the K-DETEK Study in 2024. The study involved 9,024 participants and demonstrated high specificity (99.8%) and sensitivity (78.1%). These findings are particularly beneficial for cancers without established screening programs, such as ovarian, liver, esophageal, pancreatic, and uterine cancers. (7)
Developing affordable, reliable solutions – Single-Cancer Early Detection (SCED) Tests
To enhance the detection sensitivity of ctDNA, especially for certain cancers like breast cancer and early-stage tumors, high-depth sequencing is often employed. However, this approach can be economically impractical for widespread screening.
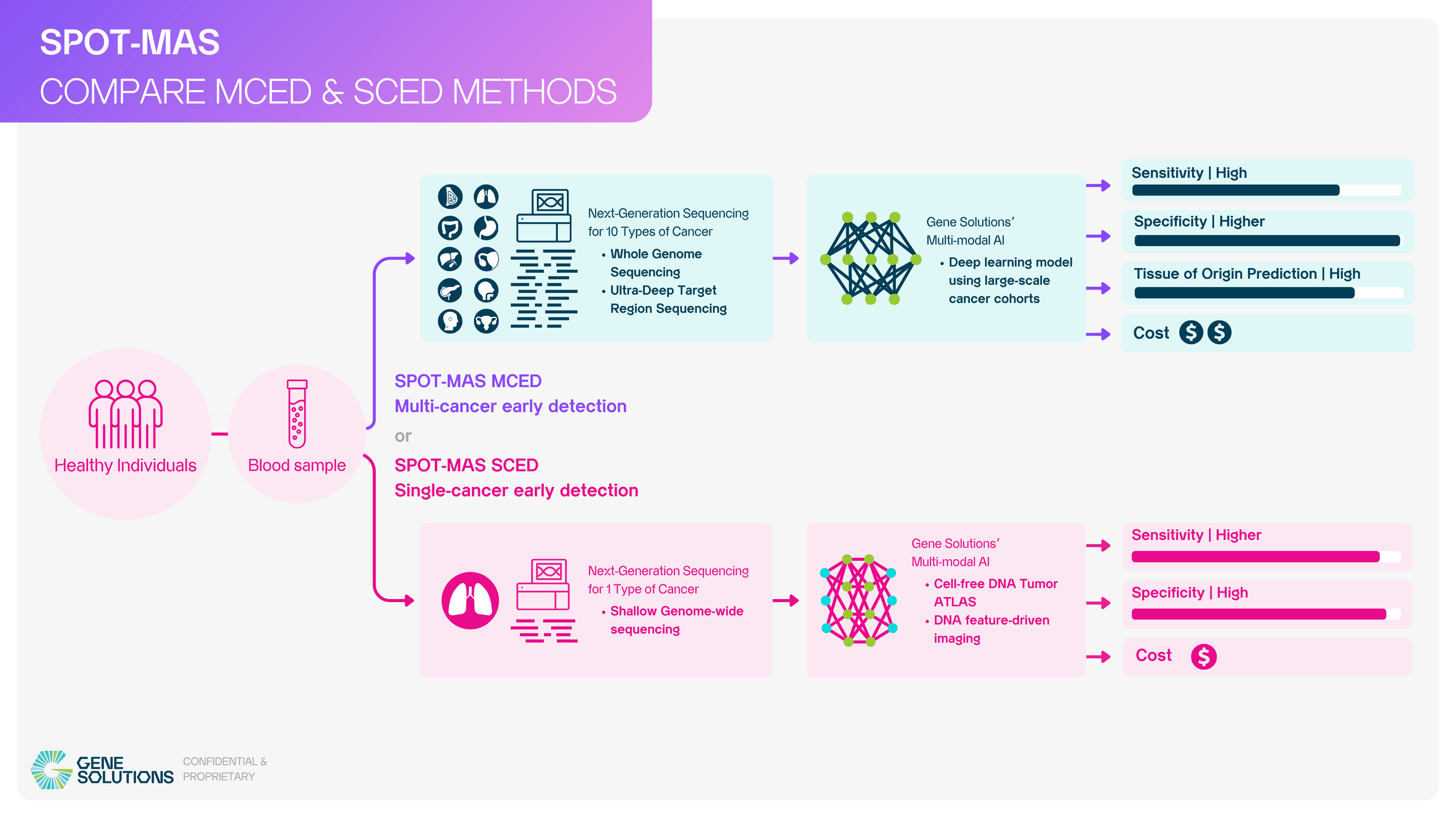
Building on the success of our validated MCED platform, Gene Solutions is now focused on expanding accessibility through Single-Cancer Early Detection (SCED) tests. This approach based on shallow genome-wide sequencing and novel feature engineering techniques leverages our advanced ctDNA technology and proprietary AI models to deliver affordable, high-accuracy cancer screening tailored to individual cancer types.
Key Advantages of the SCED Tests:
- Reduced Cost: By concentrating sequencing efforts on specific cancer characteristics, the SCED test minimizes resource requirements, significantly lowering the cost of screening.
- Enhanced Sensitivity and Specificity: Our AI models, developed using large genomic insights, improve the detection capabilities of ctDNA while reducing false positives, ensuring reliable results.
- Scalable Solution: The SCED test is designed for scalability, enabling widespread implementation across regions with varying levels of healthcare infrastructure.
At Gene Solutions, our mission is clear—to lead the way in innovative, accessible cancer screening solutions. With the successful validation of our MCED platform and the development of SCED, we are committed to making early cancer detection a reality for all.
Reference:
- Huang, J., Ngai, C. H., Deng, Y., Tin, M. S., Lok, V., Zhang, L., Yuan, J., Xu, W., Zheng, Z. J., & Wong, M. C. S. (2022). Cancer incidence and mortality in Asian countries: A trend analysis. Cancer Control: Journal of the Moffitt Cancer Center, 29, 10732748221095955. https://doi.org/10.1177/10732748221095955
- Hollingsworth, A. B. (2019). Redefining the sensitivity of screening mammography: A review. The American Journal of Surgery, 218(2), 411–418. https://doi.org/10.1016/j.amjsurg.2019.01.039
- Pinsky, P. F., Bellinger, C. R., & Miller, D. P. (2017). False-positive screens and lung cancer risk in the National Lung Screening Trial: Implications for shared decision-making. Journal of Medical Screening, 25(2), 110–112. https://doi.org/10.1177/0969141317727771
- Jones, R. M., Devers, K. J., Kuzel, A. J., & Woolf, S. H. (2010). Patient-Reported Barriers to Colorectal Cancer Screening. American Journal of Preventive Medicine, 38(5), 508–516. https://doi.org/10.1016/j.amepre.2010.01.021
- Barry, M. J., & Simmons, L. H. (2017). Prevention of Prostate Cancer Morbidity and Mortality: Primary Prevention and Early Detection. The Medical clinics of North America, 101(4), 787–806. https://doi.org/10.1016/j.mcna.2017.03.009
- Mallet, M. (2024, August 6). FDA approves blood test to screen for colon cancer. Cancer Research Institute. https://www.cancerresearch.org/blog/august-2024/fda-aproved-blood-test-to-screen-for-colon-cancer-gives-hope-to-immunologists-and-patients
- Carbonell, C., Hutchinson, J. M., Hilsden, R. J., Yang, H., & Brenner, D. R. (2024). Blood-Based Multi-Cancer Early Detection Tests (MCEDs) as a potential approach to address current gaps in cancer screening. Cancer Control, 31. https://doi.org/10.1177/10732748241307360
