Gene Solutions’ HRD Insight Expands PARP Inhibitor Access for Ovarian Cancer Patients
First-of-its-kind study reveals high HRD prevalence in Vietnamese ovarian cancer patients and validates the performance of HRD Insight assay.
Gene Solutions today announced the publication of a peer-reviewed study in Frontier in Oncology validating the company’s HRD Insight assay, a novel protocol design to detect recombination deficiency (HRD) in ovarian cancer. The study demonstrates the assay’s ability to deliver accurate and robust HRD results, even in challenging clinical samples, and underscores its potential to expand access to precision therapies globally.
HRD is a critical biomarker in ovarian cancer, used to identify patients most likely to benefit from targeted treatments such as PARP inhibitors (PARPi). While international guidelines (e.g. ESMO, NCCN) recommend HRD testing for all ovarian cancer/fallopian cancer/primary peritoneal cancer patients, access remains limited in many developing countries due to cost, complexity, and sample quality issues.
The newly published study, “Combination of BRCA deep targeted sequencing and shallow whole-genome sequencing to detect homologous recombination deficiency in ovarian cancer,” is the first to characterize HRD prevalence in Vietnamese patients and provides strong evidence supporting a simplified, cost-effective approach to testing.
Key takeaways:
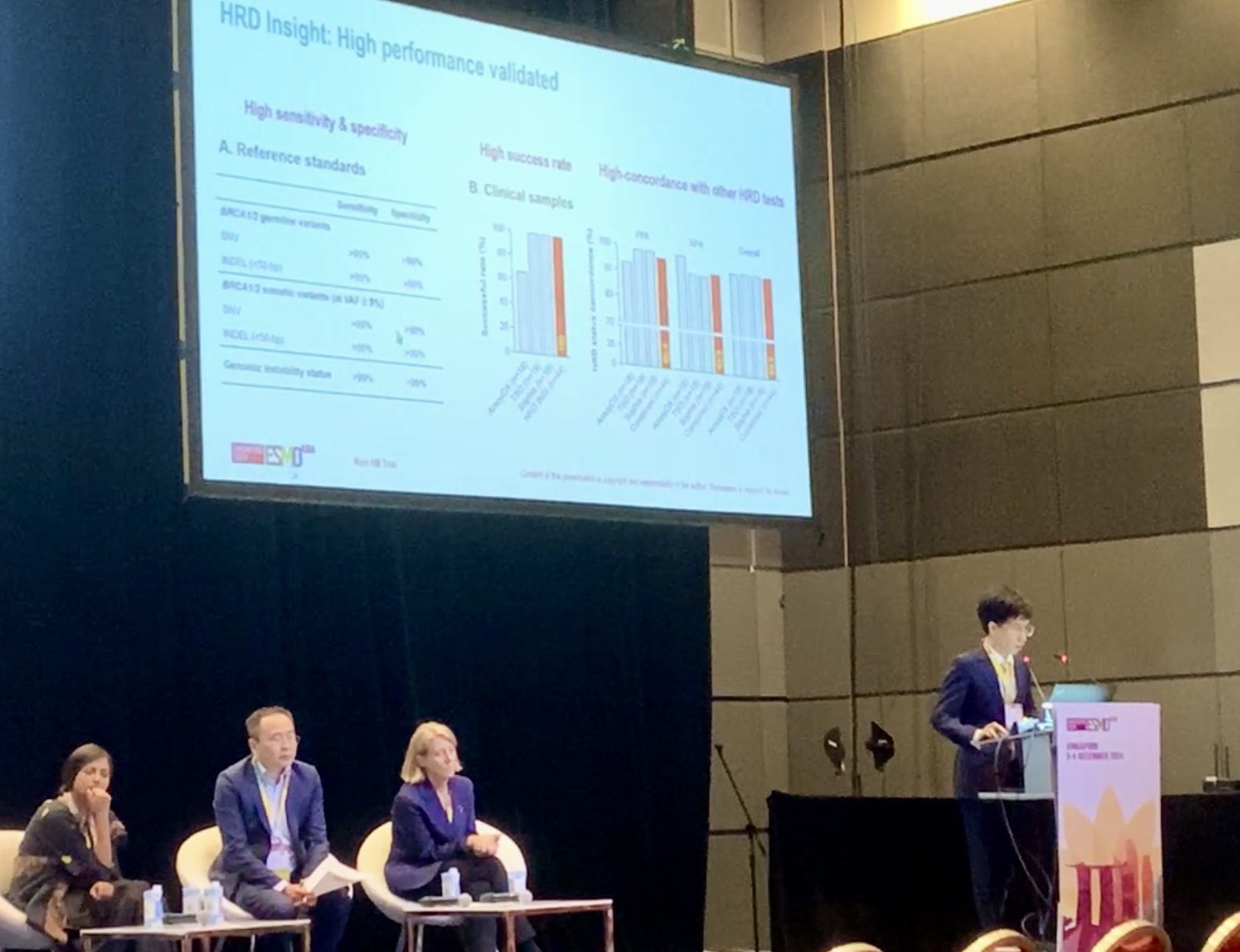
- High technical success: 94.8% sequencing success rate, even in low-quality FFPE tissue samples.
- High concordance with other ESMO-recommended HRD tests: 96.3% overall agreement; 100% positive percent agreement (PPA); 92.9% negative percent agreement (NPA).
- Meaningful clinical insight: In a cohort of 73 ovarian cancer cases, 54.8% were HRD-positive, including 40.5% of BRCA-wildtype patients identified through genomic instability scoring. This reinforces the importance of assessing genomic instability beyond BRCA1/2 mutations alone.
Read the Full Study: The article is available in Frontiers in Oncology:
Clinical Significance
At an oral presentation session ESMO Asia 2024, a summary from the Friends of Cancer Research HRD Harmonization Project emphasized the need for validated and more affordable HRD assays – an urgent but challenging task. Gene Solutions’ validation data indicates that HRD Insight is a promising step toward meeting this need.
Gene Solutions presented preliminary data on the HRD Insight assay during an oral presentation session at ESMO Asia 2024
HRD Insight assay combines deep BRCA1/2 sequencing with shallow whole-genome sequencing (sWGS) to derive a whole-genome instability (wGI) score. This integrated method delivers a comprehensive view of HRD status while remaining affordable and practical for laboratories in resource-limited settings.
“Comprehensive HRD assessment ensures that no patient misses their opportunity for PARPi treatment,” said Dr. Duy Sinh Nguyen, Oncology Medical Director at Gene Solutions. “Our findings highlight how HRD Insight can bring reliable testing to more patients and support oncologists in making informed treatment decisions.”
By validating HRD Insight against leading commercial assays and demonstrating its effectiveness in real-world clinical samples, the study positions the assay as a practical solution to expand PARPi access and improve outcomes for ovarian cancer patients.
Advancing Precision Oncology
HRD Insight is now integrated into K-4CARE™— Gene Solutions’ all-in-one platform that uniquely combines a 504-gene DNA panel, 19,435-gene mRNA-seq profiling and shallow whole-genome sequencing (sWGS) with ctDNA-MRD monitoring. This integration significantly enhances clinical decision-making by delivering:
- Comprehensive biomarker coverage: Actionable and resistance mutations (with RNA-based analysis increasing gene-fusion detection by ~25%), germline mutations, MSI, TMB, oncovirus profiling, and HRD status—supporting immunotherapy, targeted therapy selection.
- Validated HRD detection: Reliable performance even with low-input or poor-quality specimens, with applicability not only in ovarian cancer but also in breast, prostate, and pancreatic cancers.
- Personalized ctDNA-MRD monitoring (Limit of detection 0.01%): Enables real-time monitoring of treatment response and emerging resistance through personalization of up to 10 mutations per patient, plus viral DNA tracking for tumor viruses such as Human Papillomavirus (HPV) or Epstein-Barr Virus (EBV).
- Tumor tissue of origin prediction using AI algorithms: Supports management of cancers of unknown primary.

K-4CARE™ sample report (for illustration only) showing comprehensive tumor profiling in a single view.
This unified approach helps resolve the inefficiencies of sequential testing and delivers a triple advantage—time, tissue, and cost efficiency. With growing adoption in clinical trials and hospitals across Asia-Pacific, K-4CARE™ is becoming a go-to solution for precision oncology in the region.
