The MCED Test experience
The experience with multi-cancer early detection tests offers a closer look at how this approach may be shaping aspects of preventive healthcare. In the context of this MCED article, we focus on the SPOT-MAS test developed by Gene Solutions as one example of a multi-cancer early detection approach. This article outlines the general test process, highlights a few illustrative patient cases, and explores the possible benefits and challenges associated with early cancer screening worldwide.

What is the MCED Test?
The Multi-Cancer Early Detection (MCED) test is a non-invasive blood-based screening method that analyzes circulating tumor DNA (ctDNA) fragments released by tumors into the bloodstream. Using next-generation sequencing and machine-learning algorithms, MCED can detect molecular signals associated with multiple cancers at early stages, when treatment is generally more effective (1, 2). Several considerations related to MCED testing include:
- Single blood draw (~10 mL): validated in the prospective K-DETEK study involving 9,024 participants (1).
- Three scientific pillars of multi-omics:
- Genetics: Detects tumor-related DNA changes and copy number alterations indicating genomic instability (1, 3)
- Epigenetics: Identifies cancer-specific DNA methylation patterns that reveal tumor origin and activity (1, 2)
- Fragmentomics: Analyzes cfDNA fragment sizes and end motifs to uncover unique fragmentation patterns of tumor DNA (1, 2)
- Broader cancer coverage: capable of screening for the common cancers covered by traditional tests, plus the other types that currently have no standard-of-care (SOC) screening (4).
- Clinical role: Designed as a complementary tool, not a substitute for established screening.
What is the MCED Test?
The MCED Test experience
When exploring the MCED test experience, patients often want to know where the service is available and how the procedure unfolds.
Example of clinics and hospitals offering MCED
In Singapore, multi-cancer early detection (MCED) tests such as SPOT-MAS by Gene Solutions have been clinically validated in Asia (1). These tests use a multi-omics sequencing approach with artificial intelligence to screen for several common and aggressive cancer types.
MCED test: step-by-step
The MCED test experience is intended to be straightforward, potentially making early cancer screening more accessible and less invasive. This MCED assay procedure begins with a consultation to understand the scope and the implications of the test, followed by a simple 10 mL blood draw. The sample is then analyzed using using next-generation sequencing (NGS) technologies, combined with the stacking ensemble machine learning model, and results are returned after 12 working days. The final step is a post-test review with a doctor to discuss findings and plan the next course of action.
- Consultation & test order: A physician evaluates the patient’s risk profile and suitability before ordering the test.
- Blood collection: A quick, routine sample collection (requires only a 10mL blood draw). at an affiliated clinic or hospital.
- Laboratory analysis: The sample is processed at Gene Solutions Genomics Pte Ltd using next-generation sequencing and AI models.
- Results reporting: The physician receives a report in about 12 working days from the time the laboratory processes the sample.
- Post-test consultation: The physician explains the test results, including whether a ctDNA cancer signal was detected or not, and advises on appropriate follow-up actions.
Price reference
When considering the MCED test in Singapore, it is important to keep in mind that costs vary depending on the provider, the test panel, and the technologies used. Because prices and insurance coverage are not standardized, patients are encouraged to contact their chosen healthcare facility directly to obtain the most accurate and up-to-date information.
Gene Solutions MCED process
To better understand the MCED Test experience, it is helpful to see how the test is conducted as a sequence of steps. From preparation to clinic procedures and finally to receiving results, each stage plays a role in shaping the overall journey.
Preparation before the test
The preparation stage is an important part of the MCED process, ensuring both accuracy and patient understanding. Before the blood draw, individuals receive pre-test counselling. In this consultation, healthcare professionals explain the test’s purpose, review medical history, confirm eligibility, and obtain physician ordering and informed consent. This helps patients make informed decisions and prepares them for a smooth testing experience (5).
According to Gene Solutions (SPOT-MAS), the MCED test is recommended for:
- Adults aged 40 and above
- Younger individuals with high-risk factors, such as:
-
- Heavy smoking: ≥20 pack-years (6)
- Heavy alcohol consumption: ≥15 drinks/week (men) or ≥8 drinks/week (women) (7)
- Hereditary cancer risk: Positive for genes like BRCA1, BRCA2, TP53 (8)
- Chronic liver disease: Hepatitis B or C infection (9)
- Family history of cancer: First-degree relatives diagnosed with cancer (10)
Clinic visit and blood collection
The next step in the MCED test process is blood collection. Once registration and identity verification are completed, patients proceed to the clinic visit for blood collection. This stage is simple but essential, as it ensures the sample is drawn and handled correctly before being processed in a licensed laboratory for analysis.
- Doctor/nurse consultation: Any remaining questions are answered and the test order is confirmed.
- Blood draw: A single venous blood sample of about 10 mL is collected.
- Laboratory transfer: The sample is securely processed and sent to the clinical laboratory.
- Analysis: Next-generation sequencing (NGS) and stacking ensemble machine learning model are applied at the laboratory.
- Results timeline: Findings are returned to the physician in about 12 working days from the time the lab receives the sample.
Laboratory analysis and technology
Once the blood sample is collected, it is transported to a clinical laboratory in Singapore for advanced processing. At this stage, SPOT-MAS applies a multi-omics workflow on circulating tumor DNA (ctDNA) together with machine-learning models to detect cancer signals and support downstream clinical decisions (5).
- Sample preparation: Plasma is separated from whole blood and preserved under laboratory quality controls.
- Sequencing & AI: Next-generation sequencing (NGS) and stacking ensemble machine learning model
- ctDNA multi-omics analysis: The assay examines complementary signal layers: Genetics, Epigenetics and Fragmentomics to identify subtle cancer signals and to predict tissue of origin.
- Quality assurance: Testing is performed in a CAP-accredited clinical laboratory, which is designed to follow established quality systems, personnel competencies, and standardized processes.
This combination of sequencing technology and AI-driven, multi-omics ctDNA analysis enables simultaneous screening across multiple cancers while remaining non-invasive; prospective validation has demonstrated high sensitivity and specificity for SPOT-MAS in real-world cohorts (1, 3).
Timeline for receiving results and interpretation
The average turnaround time is 12 working days from the date the laboratory receives the specimen. This MCED test results timeline reflects the period needed for advanced sequencing and analysis. Once the process is completed, the results are delivered to the ordering physician and classified into one of three categories (5).
| Result Category | ctDNA Z-score | Interpretation & Next Step |
| NEGATIVE | Z-score < 2 | Interpretation:
No signals have been found to suggest the possibility that you are having any of the 10 types of cancer in the scope of investigation (Lung, Liver-Biliary tract, Breast, Colorectum, Stomach, Esophagus, Pancreas, Head & Neck, Ovary, Endometrium). Next Step: The patient should continue with existing cancer screening regimens and repeat the SPOT-MAS test annually. |
| INTERMEDIATE | 2 < Z-score < 3 | Interpretation:
There is an intermediate signal in the subthreshold range to a positive level. Next Step: The patient is strongly recommended to repeat this test within six months, free of charge. |
| POSITIVE | Z-score > 3 | Interpretation:
A detected ctDNA signal indicates that you may have cancer. This test is a screening tool, not a diagnostics test. Next Step: Your doctor will advise you on the next steps and recommend further diagnostic tests to evaluate your condition. |
Case study: MCED Test experience of patient
The following real-world MCED Test experience case studies from Gene Solutions highlight how the SPOT-MAS™ Multi-Cancer Early Detection (MCED) test has been clinically applied to uncover cancers in asymptomatic individuals. These cases demonstrate how Gene Solutions’ advanced technology enables timely medical intervention, and improved patient outcomes across multiple cancer types.
Case 1: Gastric Cancer detected in a 64-year-old
This case illustrates the effectiveness of the SPOT-MAS™ Multi-Cancer Early Detection (MCED) test in identifying common and aggressive cancers at an early stage.
First Patient Profile and Initial Screening
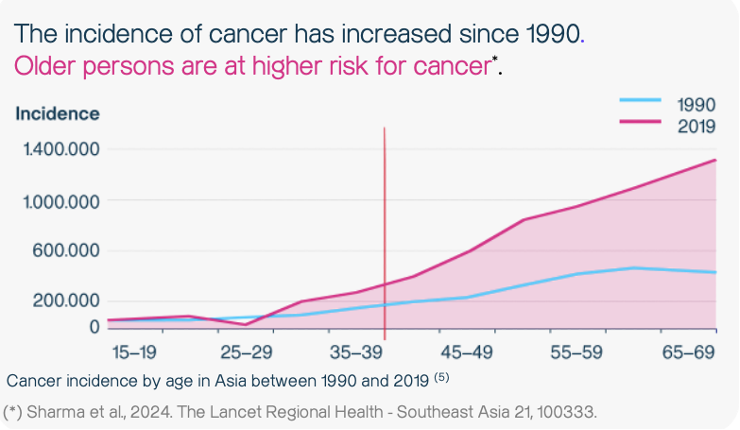
The patient was a 64-year-old male who underwent the SPOT-MAS™ test as part of a routine health screening. He had no prior history of cancer and no known family history of the disease. However, individuals over 40 years of age are generally recommended for the SPOT-MAS test, as older persons have a higher risk of developing cancer (11).
SPOT-MAS result and diagnostic pathway
- SPOT-MAS Result: The test detected a positive ctDNA signal, indicating the possible presence of tumor-derived DNA fragments in the blood.
- Tumor Origin Prediction: The SPOT-MAS test predicted potential tumour of origins in the stomach and lung.
- Confirmatory Diagnostic Steps: Following the positive result, the patient was referred for diagnostic imaging. He underwent endoscopy, which is the recommended procedure when a stomach-origin signal is detected.
Diagnosis: The endoscopy revealed suspected lesions in the antrum. A biopsy confirmed the presence of poorly differentiated signet-ring cell adenocarcinoma. A subsequent CT scan revealed enhancement in the pyloric antrum, mild fatty infiltration, and gastric stagnation, confirming the diagnosis. - Disease Staging: The patient underwent a total gastrectomy with D2 lymph node dissection, and post-surgical pathology confirmed lymph node metastasis, showing that the cancer had already spread beyond the primary tumor site.
Conclusion for case 1
This case highlights the silent and aggressive nature of gastric cancer, which often progresses without early symptoms. The ctDNA signal detected through the SPOT-MAS test enabled early medical intervention and guided timely diagnostic follow-up. Gastric (stomach) cancer is among the 10 cancer types covered by the comprehensive SPOT-MAS 10 test, emphasizing its value in broad-spectrum early detection.
Case 2: Colorectal Cancer detected in an 80-year-old
This second case demonstrates how the SPOT-MAS™ Multi-Cancer Early Detection (MCED) test can identify early-stage colorectal cancer in asymptomatic elderly individuals, offering an alternative to invasive colonoscopy screening:
Second patient Profile and Initial Screening
The patient was an 80-year-old female who underwent the SPOT-MAS™ test despite having no family history of cancer and no presenting symptoms. She belonged to the age group recommended for annual cancer screening, as colorectal cancer risk increases significantly after the age of 50 (11).
SPOT-MAS Result and Diagnostic Pathway
- SPOT-MAS Result: The test detected a positive ctDNA signal, suggesting the presence of tumor-derived DNA in the bloodstream.
- Tumor Origin Prediction: The test predicted a potential tumor signal originating from the colorectal region.
- Confirmatory Diagnostic Steps: Based on the result, the patient was advised to undergo a colonoscopy, the recommended follow-up procedure when colorectal origin is detected.
- Diagnosis: The colonoscopy identified a 2.5 cm polyp in the sigmoid colon, which was removed through polypectomy. Additionally, a 3 cm ulcerated mass was discovered in the ascending colon. Histopathology confirmed stage II adenocarcinoma.
- Treatment: The patient subsequently underwent a laparoscopic colectomy, successfully removing both the cancerous and precancerous lesions.
Conclusion for case 2
This case underscores the potential of the SPOT-MAS test to detect colorectal cancer at a stage when curative treatment is still possible. For elderly individuals who may not be suitable candidates for frequent colonoscopy, a non-invasive, blood-based test like SPOT-MAS offers a convenient and accurate screening alternative. Colorectal cancer is one of the 10 major cancer types covered by the SPOT-MAS 10 test, reinforcing its role in comprehensive early detection.
Case 3: Breast Cancer detected in a 54-year-old
This case study demonstrates the effectiveness of the SPOT-MAS™ Multi-Cancer Early Detection (MCED) test in identifying cancer at an early stage in an asymptomatic individual, enabling timely diagnosis and intervention.
Third Patient Profile and Initial Screening
The patient was a 54-year-old female who underwent the SPOT-MAS™ test. She had no previous history of cancer and no known family history of the disease (11).
SPOT-MAS Result and Prediction
The SPOT-MAS test returned a positive ctDNA signal, indicating the presence of tumor-derived DNA fragments in the blood. The test predicted the potential tumor origins in the breast and colorectum. Breast cancer is one of the ten cancer types covered under the SPOT-MAS 10 portfolio.
Confirmatory Diagnosis and Staging
- Follow-up Recommendations: After receiving a positive result, the patient was advised to undergo diagnostic imaging tests, including breast ultrasound, mammography, and colonoscopy. For a predicted breast tumor origin, the recommended diagnostic imaging is breast ultrasound combined with mammography or breast MRI.
- Imaging Findings: The mammography revealed a 23 × 33 mm lesion with poor echogenicity in the breast.
- Final Diagnosis: A biopsy was performed and confirmed invasive malignant mammary cells in fibrous tissue, with hyperchromatic and deformed nuclei, consistent with aggressive breast cancer.
- Staging: The findings were consistent with Stage IB breast cancer.
Treatment and Clinical Significance
The patient successfully underwent radical surgical treatment, removing the tumor completely. This timely intervention, initiated by the detection of a ctDNA signal, demonstrates the benefits of multi-cancer early detection in guiding early and effective clinical action. The findings emphasize the importance of regular check-ups and cancer screenings, as early detection significantly improves the 5-year survival rate for breast cancer.
Benefits and challenges
| Dimension | Key Benefits | Key Challenges |
| Clinical performance | Broad multi-cancer coverage in a single test; high specificity minimizes unnecessary follow-ups; complements existing organ-specific screening. | Positive results still require confirmatory diagnostics; limited sensitivity for some early or out-of-scope tumors. |
| Patient experience | Non-invasive single blood draw; suitable for asymptomatic adults and those reluctant to undergo invasive procedures. | Anxiety around positive or intermediate results; misunderstanding that screening equals diagnosis. |
| Operational workflow | Simple specimen collection at clinics; centralized lab analysis ensures consistency and quality; predictable turnaround times. | Clinic staff training, labeling, logistics, and cold-chain/time-to-lab coordination; scheduling follow-ups. |
| Health-system impact | Earlier detection can shift stage at diagnosis and potentially reduce long-term treatment burden; fills gaps where no routine screening exists. | Added demand on imaging/endoscopy for confirmatory work-ups; capacity bottlenecks if uptake is high. |
| Economics | One test screens for multiple cancers, potentially improving cost-effectiveness versus multiple single-organ screens. | Upfront test cost and downstream diagnostics can be significant for self-pay patients. |
| Technology & data | Multi-omics plus AI improves signal detection and tumor-of-origin prediction; continuous model improvement with growing datasets. | AI models need ongoing validation and governance; rare cancer signals may be harder to localize. |
Frequently asked questions
To help patients better understand the MCED Test Singapore experience, below are some of the most common questions about the testing process, comfort level, comparison with conventional screenings, and why many individuals choose Gene Solutions for their early detection journey.
How long does it take to get results?
Results are typically available within 12 working days, depending on the testing center. Once processed, the results are delivered to the ordering physician, who will discuss them with the patient (1).
Is the MCED test painful or invasive?
The SPOT-MAS test requires only a single venous blood draw of 10 mL, performed by trained medical staff. The procedure takes just a few minutes and does not require fasting or any special preparation.
Can the MCED test replace traditional cancer screenings?
No. The MCED test is designed to complement, not replace existing national screening programs (e.g., mammography, colonoscopy, or LDCT). A positive MCED result still requires confirmatory diagnostic procedures according to clinical guidelines.
Why choose Gene Solutions for the MCED test?
Gene Solutions developed Asia’s first clinically validated MCED test, supported by a prospective cohort of 9,024 asymptomatic participants published in BMC Medicine (1). The company operates CAP-accredited laboratories in Asia and applies proprietary multi-omics plus AI technology (DNAsphere.AI) for signal detection and tumor-of-origin prediction. To date, the SPOT-MAS platform is supported by 19 peer-reviewed publications and real-world evidence across Southeast Asia (5).
Conclusion
The MCED Test Singapore experience represents an important step forward in precision and preventive healthcare. Through the combination of advanced technology, the SPOT MAS test by Gene Solutions allows the early detection of multiple cancer types in their silent stages with only a single blood sample. Real world case studies in Singapore have shown that this method can uncover cancers, support timely treatment, and improve patient outcomes.
Although traditional screenings remain essential, the MCED test serves as a complementary approach that broadens early detection to include cancers not covered by standard programs. With Singapore’s increasing focus on proactive health management, Gene Solutions and its clinically validated technology provide both patients and healthcare professionals with an accessible and evidence based solution to identify cancer early, when treatment is most effective.
Disclaimer: This content is for educational purposes only and does not constitute medical advice. Multi-Cancer Early Detection (MCED) tests should be considered and ordered by licensed healthcare professionals. Availability and regulatory permissions vary by country.
Reference:
(1) Nguyen, L. H. D., et al. “Prospective validation study: a non-invasive circulating tumor DNA-based assay for simultaneous early detection of multiple cancers in asymptomatic adults (SPOT-MAS).” BMC Medicine 23, 90 (2025). https://doi.org/10.1186/s12916-025-03929-y
(2) Nguyen, T. H., et al. “Multimodal analysis of methylomics and fragmentomics in plasma cell-free DNA for multi-cancer early detection and localization (SPOT-MAS).” Frontiers in Genetics 14, 1158121 (2023). https://doi.org/10.3389/fgene.2023.1158121
(3) Nguyen, T. H. H., et al. “Combination of Hotspot Mutations With Methylation and SPOT-MAS Predictions Improves Early-Stage Cancer Detection.” Cancers (Basel) 17, 604 (2025). https://doi.org/10.3390/cancers17040604
(4) WHO, American Cancer Society 2024 and USPSTF recommendations
(5) Gene Solutions. SPOT-MAS Patients – Multi-Cancer Early Detection Test. Gene Solutions, 2025. Retrieved 26 October 2025, from https://spotmas.com/patients/
(6) Chen et al., 2021. American Journal of Otolaryngology 42, 102915
(7) Connor et al., 2017. Addiction 112, 222–228
(8) Garber et al., 2005. JCO 23, 276–292
(9) Petruzziello et al., 2018. TOVJ 12, 26–32
(10) Brewer et al., 2017. Breast Cancer Res Treat 165, 193–200
(11) Luu Hong Dang Nguyen et al. 2024. A consultation and work-up diagnosis protocol for a multicancer early detection test: a case series study. Future Science OA, 10(1) https://doi.org/10.1080/20565623.2024.2395244
