A guide to Multi-Cancer Early Detection Testing (MCED) in Asia

MCED testing is emerging as an innovative solution in the fight against cancer, offering the ability to detect multiple types of cancer at earlier stages with just a single test. This approach is transforming preventive healthcare globally, giving people a better chance for timely treatment and improved outcomes. Let’s explore the guide to Multi-Cancer Early Detection Testing, particularly in Asia!
What is MCED?
Multi-Cancer Early Detection (MCED) testing is an emerging approach in cancer prevention that uses a single blood sample to screen for multiple cancer types—often before symptoms appear. By identifying molecular signals shed by tumors into the bloodstream, MCED aims to support earlier diagnosis, timely treatment, and improved outcomes.
Unlike traditional screening methods that focus on one cancer at a time, MCED offers a broader scope and a simpler patient experience, potentially filling gaps in current preventive care.
Defining MCED
MCED refers to blood-based tests that analyze circulating tumor DNA (ctDNA) features associated with cancer. These tests do not diagnose cancer directly but estimate the likelihood of a cancer signal and may suggest a probable tissue of origin, guiding further diagnostic steps.
Evolution of MCED Testing
The idea of detecting cancer through blood traces has existed for decades. However, only recent advances in molecular biology, next-generation sequencing, and computational modeling have made multi-cancer screening feasible at scale.
One such innovation is SPOT-MAS™, developed by Gene Solutions. It is a clinically validated MCED test that integrates genetic, epigenetics, fragmentomics patterns from ctDNA, analyzed using proprietary AI algorithms. SPOT-MAS has been evaluated in over 9,000 participants across Asia and is recognized for its cost-effective, accredited testing model (1).
Note: SPOT-MAS is intended for use under medical supervision and is not available for direct consumer ordering.
What is MCED?
MCED vs Traditional Cancer Screening
| Aspect | MCED (Multi-Cancer Early Detection) –
SPOT-MAS |
Traditional Screening |
| Scope of detection | Screens for signals from 10 major cancers and 75 subtypes in one blood test (2). | Focuses on one cancer at a time (e.g., mammogram, colonoscopy). |
| Markers analyzed | ctDNA features interpreted by computational algorithms. | Specific proteins or imaging findings tied to individual cancers. |
| Availability | Emerging in clinical practice; adoption varies by region. | Widely established for select cancers (e.g., breast, lung, colorectal). |
| Detection of cancers without standard screening | Yes — includes cancers like liver–biliary tract, stomach, esophagus, ovary, pancreas, endometrium, head & neck. | No — limited to cancers with long-standing screening programs. |
| Patient experience | Minimally invasive — single blood draw. | May involve invasive or uncomfortable procedures. |
| Integration with care | Complements existing methods; guides follow-up diagnostics. | Functions independently for specific cancers. |
MCED is not a replacement for traditional screening. It is designed to work alongside established programs, offering broader coverage and supporting earlier detection where standard protocols may not exist.
MCED Workflow: From Blood Draw to Result
Understanding the Clinical Pathway
A practical way to understand MCED is to follow the patient journey, from initial consultation to follow-up diagnostics. Each step ensures the test is used appropriately and results are clinically meaningful.
Step-by-Step Workflow
- Pre-test consultation
A physician explains the purpose of MCED, its accuracy, limitations, and what a positive, intermediate, or negative result means. Informed consent is obtained. - Blood draw
About 10 ml of blood is collected via venipuncture. The process is quick and minimally invasive. - Sample processing
Plasma is separated, and cell-free DNA (cfDNA) is extracted for analysis. - Sequencing & analysis
Next-generation sequencing (NGS) reads genetic, epigenetics, fragmentomics patterns - Algorithmic classification
AI models interpret these signals to estimate the likelihood of cancer and predict the probable tissue of origin (TOO). - Post-test consultation
The physician reviews results. If positive, confirmatory diagnostic imaging will identify the lesion, followed by biopsy if indicated.
 How MCED Technology Works
How MCED Technology Works
How MCED Technology Works
MCED uses liquid biopsy technology, analyzing ctDNA—tumor-derived fragments circulating in blood. Unlike traditional tumor markers that focus on single proteins, MCED applies multimodal analysis for higher specificity and sensitivity.
Key components:
- Genetic: Detects tumor-related DNA changes and copy number alterations indicating genomic instability.
- Epigenetics: Identifies cancer-specific DNA methylation patterns that reveal tumor origin and activity.
- Fragmentomics: Analyzes cfDNA fragment sizes and end motifs to uncover unique fragmentation patterns of tumor DNA.
- AI modeling: Machine learning algorithms trained on large datasets interpret complex signals.
Limitations and Ongoing Challenges
- Variable sensitivity by cancer type and stage
Detection rates differ. For example, colorectal cancer shows high sensitivity, while early-stage breast cancer has historically been harder to detect. Multi-omics approaches like SPOT-MAS have improved performance in distinguishing early-stage breast cancer from benign lesions (3). - Confirmatory diagnostics required
A positive MCED result is not diagnostic. Imaging and biopsy remain essential. - False positives and negatives
Rare but possible. False negatives occur when ctDNA shedding is minimal.

Limitations and Ongoing Challenges
MCED Test Accuracy & Performance
Sensitivity by Cancer Type
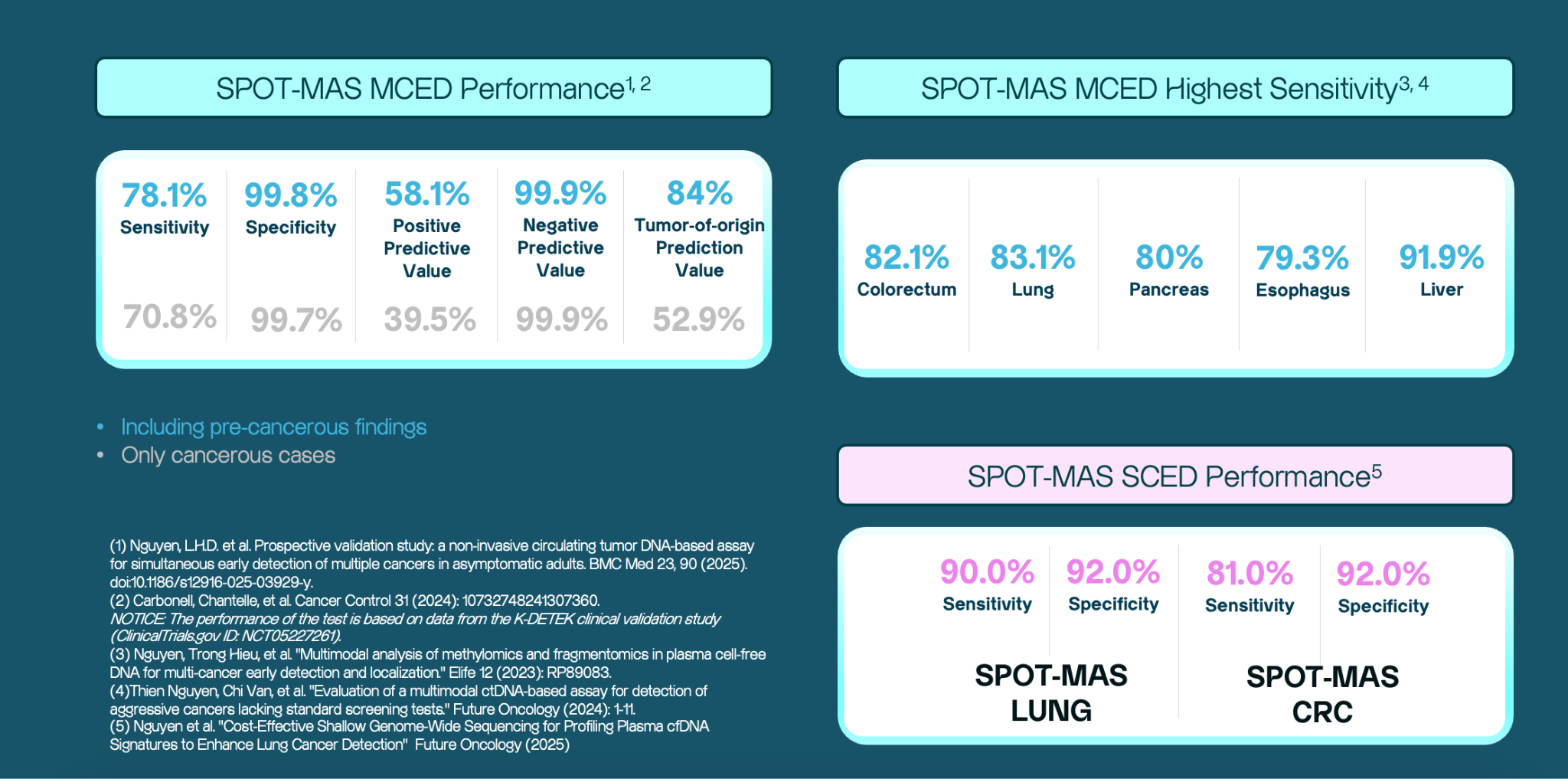
MCED sensitivity varies (1, 4):
- Overall sensitivity: 78.1%
- Colorectum: 82.1%
- Lung: 83.1%
- Pancreas: 80%
- Esophagus: 79.3%
- Liver: 91.9%
Clinical Trial Evidence
- K-DETEK (9,024 participants): Asia’s first and largest prospective validation of MCED (SPOT-MAS), demonstrating specificity and strong potential for clinical integration.
Why Early, Multi-Cancer Detection Matters
Most cancer-related mortality occur in cancers without routine screening programs. Detecting these cancers early dramatically improves survival.
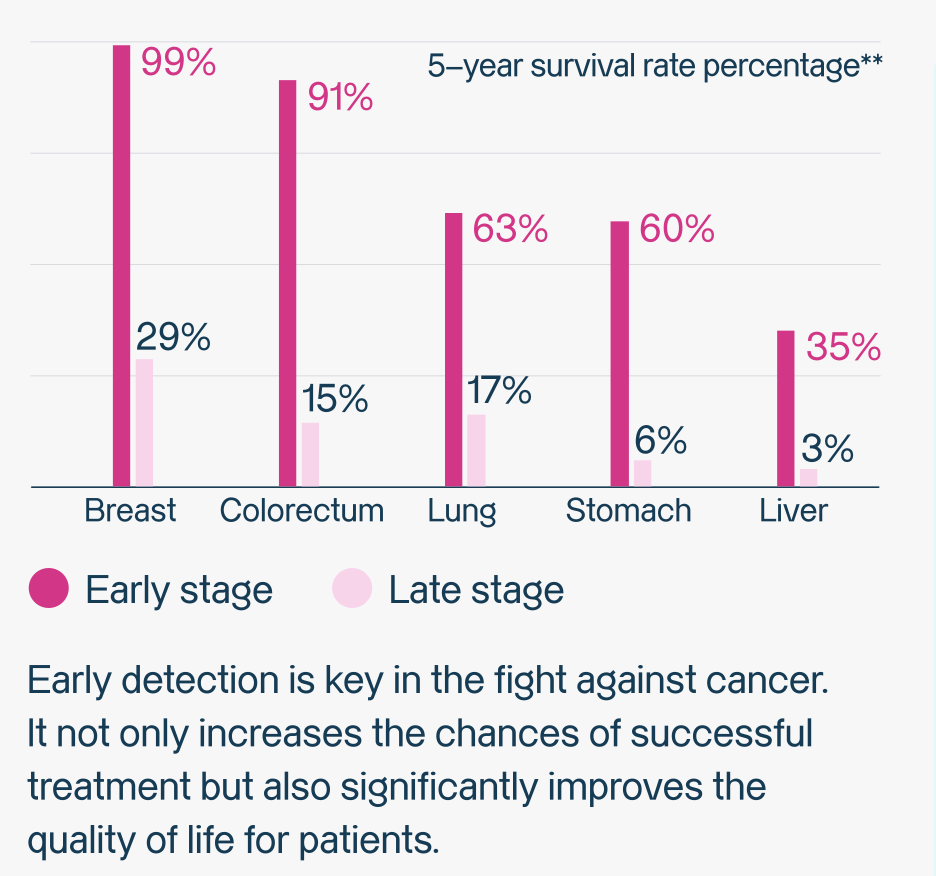
Survival Impact Examples
Why Early, Multi-Cancer Detection Matters?
- Breast cancer
Early stage: >99% 5-year survival
Advanced stage: 29% survival
→ Early detection improves outcomes by 70 percentage points. - Liver cancer
Early stage: ~35% survival
Advanced stage: ~3% survival
→ Early detection increases survival odds 11-fold.
Why MCED matters: A single blood draw can screen for multiple cancers, potentially shifting diagnosis from late-stage, hard-to-treat disease to early-stage, more treatable conditions (5).

Why is Early, Multi-Cancer Detection a Game-Changer?
Who Should Consider MCED Testing?
Identifying Suitable Candidates
MCED is not intended for everyone. It may be most valuable for individuals at higher risk of developing cancer, where broader detection could improve outcomes.
Ideal Candidates
MCED testing may be considered for:
- Adults aged 40 and above
- Younger individuals with high-risk factors, such as:
- Heavy smoking: ≥20 pack-years (6)
- Heavy alcohol consumption: ≥15 drinks/week (men) or ≥8 drinks/week (women) (7)
- Hereditary cancer risk: Positive for genes like BRCA1, BRCA2, TP53 (8)
- Chronic liver disease: Hepatitis B or C infection (9)
- Family history of cancer: First-degree relatives diagnosed with cancer (10)
Current Limitations
- Complementary role: MCED should not replace standard screenings (e.g., mammogram, colonoscopy).
- Insurance coverage: Limited in most regions; out-of-pocket costs may apply.
- Cost considerations: Generally higher than single-cancer screening methods.

Who Should Consider MCED Testing?
MCED Test Cost & Process
What to Expect in Singapore
Singapore is among Asia’s leaders in MCED adoption, supported by advanced healthcare infrastructure and internationally accredited laboratories.
Average cost
Pricing varies by provider, the scope of cancer coverage, and the test package. Patients should consult their healthcare provider for an accurate cost estimate.
Where to Access MCED Testing
MCED services are available through licensed hospitals, clinics, and genetic testing providers. For example:
MCED Test in Singapore
- Advantage Medical Group
- Asiamedic Wellness Assessment Centre
- ATA Medical Clinic
- Bethesda Medical
- CHI Longevity
- Complete Healthcare International Pte. Ltd.
- Crawfurd Hospital & Medical Centre
- DrAnywhere
- Fullerton Group
- HealthConnexion Medical Clinic
- Healthway Group
- HMI Medical Centre
- Minmed Group
- Pinnacle Family Clinic
- Precious Medical Centre
- Raffles Hospital
(Always confirm availability and physician consultation requirements.)
MCED Test in Malaysia
- Sunway Medical Centre
- Sunway Medical Centre Velocity
- Beacon Hospital
- Pantai Hospital Kuala Lumpur
- Prince Court Medical Centre
- National Cancer Society Malaysia
- Thomson Medical Centre
- Pantai Hospital Melaka
- Mahkota Medical Centre
- Regency Specialist Hospital
- Regency Medical Care Centre
- Blessono Specialist Clinic
MCED tests are also available in more regions across Asia, including India, Taiwan, Hong Kong, Thailand, Vietnam, Indonesia, and the Philippines.
Note: Availability and regulations differ by country. Always verify local guidelines before scheduling.

Where to get the MCED Test?
Step-by-Step Testing Process (Singapore Example)
Patients undergoing MCED in Singapore can expect a streamlined process that follows international standards, ensuring both accuracy and convenience.
- Appointment booking via hotline, clinic website, or hospital app
- Pre-test consultation: Physician explains accuracy, limitations, and next steps
- Blood draw: ~10 ml sample collected in ~10 minutes
- Laboratory analysis: Performed in accredited labs using advanced sequencing
- Results report: Delivered within ~12 working days, including Tissue of Origin (TOO) prediction
- Post-test consultation: Physician recommends diagnosis process if cancer signals are detected
Future of MCED Technology
What’s Next for Multi-Cancer Early Detection?
MCED testing is evolving rapidly, driven by advances in molecular biology, AI-powered analytics, and growing healthcare adoption. These developments aim to make cancer detection earlier, more accurate, and more accessible.
Technology Improvements
- Enhanced Tissue-of-Origin Prediction
Recent studies report up to 84% accuracy in predicting the likely tissue of origin (TOO), helping clinicians plan targeted diagnostic imaging and treatment faster. - AI and Machine Learning Integration
Advanced algorithms improve signal interpretation, reducing false positives and false negatives, and supporting better clinical decision-making.
Preventive Care
Global healthcare systems are beginning to explore MCED integration into preventive care frameworks. Key drivers include:
- Regulatory progress: Large-scale trials like K-DETEK provide evidence for clinical utility.
- Insurance adoption: Early discussions are underway to include MCED in coverage plans.
- Corporate wellness programs: Employers are considering MCED as part of proactive health initiatives.
Frequently Asked Questions
Q: Can MCED replace colonoscopy?
No. MCED is designed to complement, not replace, standard screenings such as colonoscopy.
Q: How accurate is MCED for early-stage cancer?
Sensitivity varies by cancer type and stage. For example, colorectal cancer shows high sensitivity, while early-stage breast cancer remains challenging but is improving with multi-omics approaches.
MCED Performance
Q: What happens if my MCED test is positive?
A positive result requires confirmatory diagnostics—typically imaging and biopsy—to verify cancer presence and guide treatment planning.
Gene Solutions and the Future of MCED
Gene Solutions is committed to making advanced multi-cancer early detection accessible and clinically reliable. Our approach combines:
- Comprehensive coverage: Detects signals from 10 major cancers and 75 subtypes in a single blood draw.
- Integration with health screening: Works alongside routine check-up programs for holistic preventive care.
- Post-test support program: Support confirmatory imaging pathways when ctDNA signals are detected (availability varies by country).
Through partnerships with leading healthcare providers and telemedicine options, Gene Solutions is expanding MCED access across Asia and beyond—helping individuals and organizations take proactive steps toward better cancer prevention.

Gene Solutions MCED Testing Services
Conclusions:
MCED enables the detection of 10 cancer types from a single blood test, helping identify aggressive cancers earlier when treatment is most effective. By complementing standard screenings, it fills critical gaps in preventive care. Gene Solutions makes this advanced technology accessible with expert support and trusted healthcare partnerships.
Disclaimer: This content is for educational purposes only and does not constitute medical advice. Multi-Cancer Early Detection (MCED) tests should be considered and ordered by licensed healthcare professionals. Availability and regulatory permissions vary by country.
Reference
(1) Nguyen, et al., 2025. “Prospective validation study: a non-invasive circulating tumor DNA-based assay for simultaneous early detection of multiple cancers in asymptomatic adults.” BMC Medicine 23, 90. https://doi.org/10.1186/s12916-025-03929-y
(2) Gene Solutions Multi-omics dataset on cancer subtypes (Accessed Sep 2025)
(3) Le Son Tran et al. 2025. Multimodal analysis of cell-free DNA enhances differentiation of early-stage breast cancer from benign lesions and healthy individuals. BMC Biology. doi: 10.1186/s12915-025-02371-z
(4) Carbonell, Chantelle, et al. Cancer Control 31 (2024): 10732748241307360
(5) WHO, American Cancer Society 2024 and USPSTF recommendations
(6) Chen et al., 2021. American Journal of Otolaryngology 42, 102915
(7) Connor et al., 2017. Addiction 112, 222–228
(8) Garber et al., 2005. JCO 23, 276–292
(9) Petruzziello et al., 2018. TOVJ 12, 26–32
(10) Brewer et al., 2017. Breast Cancer Res Treat 165, 193–200
