Asia’s First Large-Scale Validated Multi-Cancer Test to Transform Early Detection in China
The 3rd Guangdong Cancer Screening and Early Diagnosis and Treatment Conference, held on June 20–21, 2025, highlighted transformative advancements in early cancer detection. Co-hosted by the Guangdong Cancer Center, the Guangdong Anti-Cancer Association’s Committee of Early Cancer Diagnosis and Treatment, and the Cancer Hospital Chinese Academy of Medical Sciences Shenzhen Center, with support from ZaoDx, the event convened national oncology leaders and global innovators to address Guangdong’s cancer burden.
Ms. Ida Deleskog Lindstroem, Global Medical Affairs Director at Gene Solutions, delivered a keynote titled:
“SPOT-MAS Multi-omics Technology: Transforming Early Cancer Detection.”
She introduced SPOT-MAS, Asia’s first clinically validated multi-cancer early detection (MCED) test in a large-scale prospective study, designed to transform cancer screening.

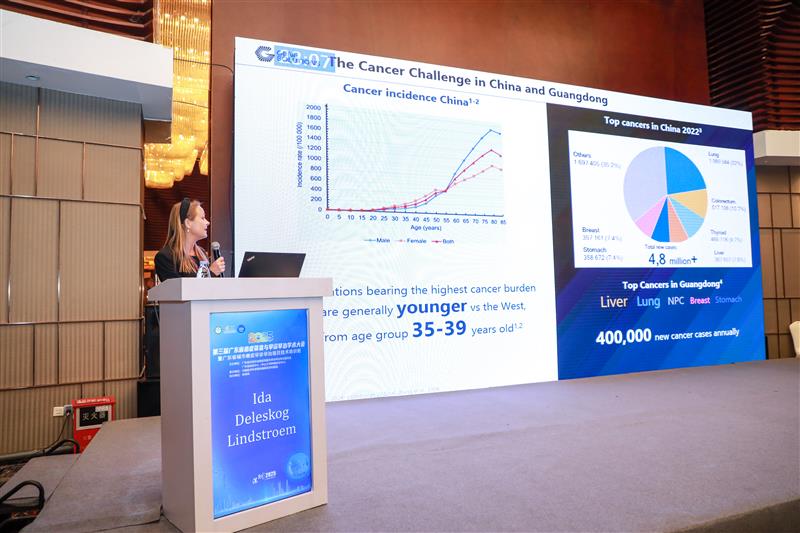
Ms. Ida outlined China’s cancer landscape, where incidence generally peaks earlier (ages 35–39) than in Western countries, significantly impacting the working-age population. In 2022, China reported 4.8 million new cases with lung, colorectal, thyroid and liver cancers being the most predominant. In Guangdong, liver, lung, and nasopharyngeal are the leading cancers, followed by breast and stomach cancers, with many currently lacking adequate screening coverage. This underscores the urgent need to implement earlier and more comprehensive cancer detection strategies.
Cancer prevention and treatment present major challenges, with early detection being critical. Early-stage diagnosis yields 80–90% survival rates compared to 10–20% for late-stage cases. Moreover, late-stage treatment cost 2–3 times more than early intervention, leading to severe economic consequences, including reduced productivity and increased national healthcare expenditures.
Amid heightened government emphasis, China’s cancer screening programs cover approximately 60% of common cancers through single-cancer screening modalities and mostly focus on high-risk populations. Yet over 40% of cancers lack screening programs, contributing to over 50% of cancer deaths.
Considering these challenges, MCED (Multi-Cancer Early Detection) technology emerges as a breakthrough solution. Ms. Ida highlighted SPOT-MAS, a next generation MCED, which integrates multi-omics with advanced artificial intelligence (AI). It targets 10 common and aggressive cancers—covering 90% of the cancers currently lacking national screening programs in China.
SPOT-MAS is supported by over 18 peer-reviewed publications and has been implemented in selected next-generation sequencing (NGS) laboratories across Asia. Using NGS and AI, SPOT-MAS analyzes circulating tumor DNA (ctDNA) shed by cancer cells into the bloodstream through multi-omics—genomic, epigenomic, and fragmentomics—enabling early signal detection of cancer. Since its launch in 2018, the SPOT-MAS platform has undergone rigorous prospective clinical validation in over 10,000 individuals, supplemented by real-world evidence from more than 10,500 cases.
In these studies, SPOT-MAS demonstrated: 78.1% sensitivity (including pre-cancerous lesions), 99.8% specificity, and 58.1% positive predictive value (PPV).
Compared to other MCED platforms such as Galleri and Shield as well as traditional tools like tumor markers, mammography, colonoscopy, and low-dose CT, SPOT-MAS has comparable or higher sensitivity, specificity and importantly a lower rate of false positives.
Ms. Ida emphasized that SPOT-MAS is not a replacement for existing standard cancer screening programs, but a powerful complementary tool—particularly for high-risk populations for cancers lacking screening options, and to improve accessibility of cancer screening to rural areas. The test is convenient, minimally invasive, affordable, and highly accessible, helping to reduce hospital congestion and free up valuable medical resources, with significant potential to ease the financial burden on patients and society.
Early cancer detection can significantly improve five-year survival rates, reduce direct treatment costs, mitigate indirect costs such as lost income and caregiver burden, delivering substantial societal benefits. SPOT-MAS could save millions of lives, alleviate the financial and emotional toll of late-stage cancer, and support China’s “Healthy China 2030” goal of reducing cancer mortality by 30%.
Ms. Ida concluded that MCED tests such as the SPOT-MAS test has the potential to transform cancer control in China, enabling early detection and offering hope to countless patients. 
References
- Cao, M., et al. (2021). Cancer screening in China: The current status, challenges, and suggestions. Cancer Letters, 506, 120–127.
- Diao, Y., et al. (2025). Cancer incidence and mortality in China, 2022. Chinese Journal of Cancer Research, 37(1), 1–12.
- Globocan. (2022). Cancer incidence and mortality statistics.
- Nguyen, L.H.D., et al. (2024). A consultation and work-up diagnosis protocol for a multicancer early detection test: A case series study. Future Sci OA, 10(1), 2395244.
- Nguyen, L.H.D., Nguyen, T.H.H., Le, V.H., et al. (2025). Prospective validation study: A non-invasive circulating DNA-based assay for simultaneous early detection of multiple cancers in asymptomatic adults. BMC Medicine, 23, 90.
- Wan, J., et al. (2025). Comparative analysis of MCED technologies in Asian populations. Journal of Precision Medicine, 8(2), 89–97.
- Xu, L., et al. (2025). Real-world evidence of SPOT-MAS in early cancer detection. Oncology Advances, 6(1), 34–42.
- Zhang, S., et al. (2024). Cancer epidemiology and screening gaps in Guangdong, China. Public Health Reports, 139(4), 456–464.
