Bridging Genomic Insights: AI‑Powered Liquid Biopsy and Multi‑Omics at ESMO Asia Congress 2025
At ESMO Asia Congress 2025 (December 5–7), Gene Solutions’ Industry Satellite Symposium brought together leading voices from across the region to examine how AI-driven liquid biopsy and multi‑omics are reshaping oncology – from early detection to MRD-guided treatment decisions and clinical utility standards.
Chaired by Prof. Herbert Ho Fung Loong (The Chinese University of Hong Kong, Hong Kong SAR) and Prof. Nick Pavlakis (The University of Sydney, Australia), the symposium – “AI-powered liquid biopsy and multi-omics in action: Bridging the gap in genomic insights across the Asia-Pacific region” – drew a full house audience. It featured actionable clinical evidence spanning early detection, treatment selection, and post-treatment monitoring.
Setting the Stage: Why AI + Liquid Biopsy Now?
Opening the session, Prof. Herbert Ho Fung Loong framed the hour around the promise of “AI‑driven multi‑omics biomarkers and liquid biopsy to transform patient outcomes”, with ambitions that span earlier detection for average‑risk individuals and precise MRD‑guided treatment escalation in early-stage disease. The faculty – Asst. Prof. Aaron C. Tan (National Cancer Centre Singapore, Singapore), Dr. Le Son Tran and Dr. Dang L.H. Nguyen (Medical Genetics Institute, Vietnam), and Prof. Aya El Helali (The University of Hong Kong, Hong Kong SAR) – shared evidence and real‑world experience that collectively argue for integrating ctDNA and AI‑enabled multi-omics analytics into routine decision‑making across the cancer continuum.

From left to right: Prof. Aya El Helali (The University of Hong Kong), Prof. Nick Pavlakis (The University of Sydney, Australia), Prof. Herbert Ho Fung Loong (The Chinese University of Hong Kong), Dr. Le Son Tran (Medical Genetics Institute, Vietnam), Asst. Prof. Aaron C. Tan (National Cancer Centre Singapore), and Dr. Dang L.H. Nguyen (Medical Genetics Institute, Vietnam)
Liquid Biopsy Across the Continuum
Speaker: Prof. Nick Pavlakis – The value of liquid biopsy at each stage of cancer care

“ctDNA can provide value at every stage of cancer care – from early detection to advanced oncology,” Prof. Pavlakis emphasized, outlining its role from diagnosis to resistance monitoring.
When using as a genomic profiling tool for cancer patients, he highlighted how liquid biopsy complements tissue-based testing, especially when 10–20% of tissue samples are inadequate for NGS, and offers faster turnaround times compared to conventional approaches.
A key advantage of ctDNA is its ability to capture spatial and temporal tumor heterogeneity, providing a more comprehensive molecular snapshot than single-site tissue biopsies. This is critical for guiding therapy in cancers with complex resistance patterns, such as EGFR-mutant NSCLC.
On ctDNA-MRD, Prof. Pavlakis highlighted prospective and real-world data of K-TRACK assay, validated across multiple tumor types including colorectal, breast, lung, HCC, and gastric cancers. Findings consistently show:
- Preoperative MRD clearance correlates with pathologic response (pCR).
- Postoperative MRD positivity predicts poor prognosis, reinforcing its role in risk stratification.
- Real-world data from Asia demonstrate that ctDNA can anticipate clinical progression up to 14 months before imaging and identify resistance mechanisms in metastatic settings.(1)
He concluded: “MRD guided decisions—escalation for ctDNA-positive or de-escalation for ctDNA-negative – are becoming central to precision oncology.”
AI-Powered Multi-Omics for Early Screening (MCED)
The team from Gene Solutions presented SPOT‑MAS, an AI-powered multi-omics liquid biopsy test designed to transform cancer screening across Asia. By integrating genetic, epigenetic, and fragmentomic signatures from a single blood draw, SPOT-MAS aims to overcome the limitations of traditional screening methods and deliver accurate, non-invasive detection of 10 cancer types and 75 subtypes.
Dr. Le Son Tran: From Innovation to Clinical Validation

Dr. Tran highlighted the technological innovation behind SPOT-MAS and its journey from analytical validation to large-scale clinical trials. Unlike conventional single-biomarker assays, SPOT-MAS employs a multi-omics approach combined with machine learning to maximize sensitivity for early-stage tumors.
The centerpiece of his talk was the K-DETEK prospective study, the third MCED clinical validation in worldwide, enrolling 9,024 asymptomatic participants. Published in BMC Medicine earlier this year, K-DETEK demonstrated 78.1% sensitivity and 99.8% specificity, outperforming other clinically validated, single-omic tests. Its proprietary AI model for tissue-of-origin (TOO) prediction was trained on data from more than 20,000 cancer patients and 25,000 healthy controls, achieving 84% TOO accuracy across retrospective (K-DISCOVERY) and prospective (K-DETEK) cohorts.
Dr. Tran summarized the mission behind SPOT-MAS with a clear statement: “For any screening tool to have a population-level impact, the test needs to be affordable. Striking the right balance between performance and accessibility is the key challenge – and SPOT-MAS was designed to meet that challenge.” He further explained how this balance is achieved: “The challenge is balancing multi-cancer early detection performance with affordability – we achieve this via robust feature engineering, supervised machine learning, and high quality multi-omics training datasets”
Dr. Dang L.H. Nguyen: The Largest Real-World Evidence Across Asia
Dr. Dang L.H. Nguyen shifted the focus to real‑world performance of SPOT-MAS in 10 cancers. In a large real-world study comprising 12,281 individuals (median age 43 years) with enrichment for familial cancer risk (24.5%), SPOT-MAS assay demonstrated a sensitivity of 78.2%, specificity of 99.8%, positive predictive value (PPV) of 67%, negative predictive value (NPV) of 99.9%, and tumor-of-origin (TOO) prediction accuracy of 80.3%. These results were comparable to previously reported in the K-DETEK prospective validation study.
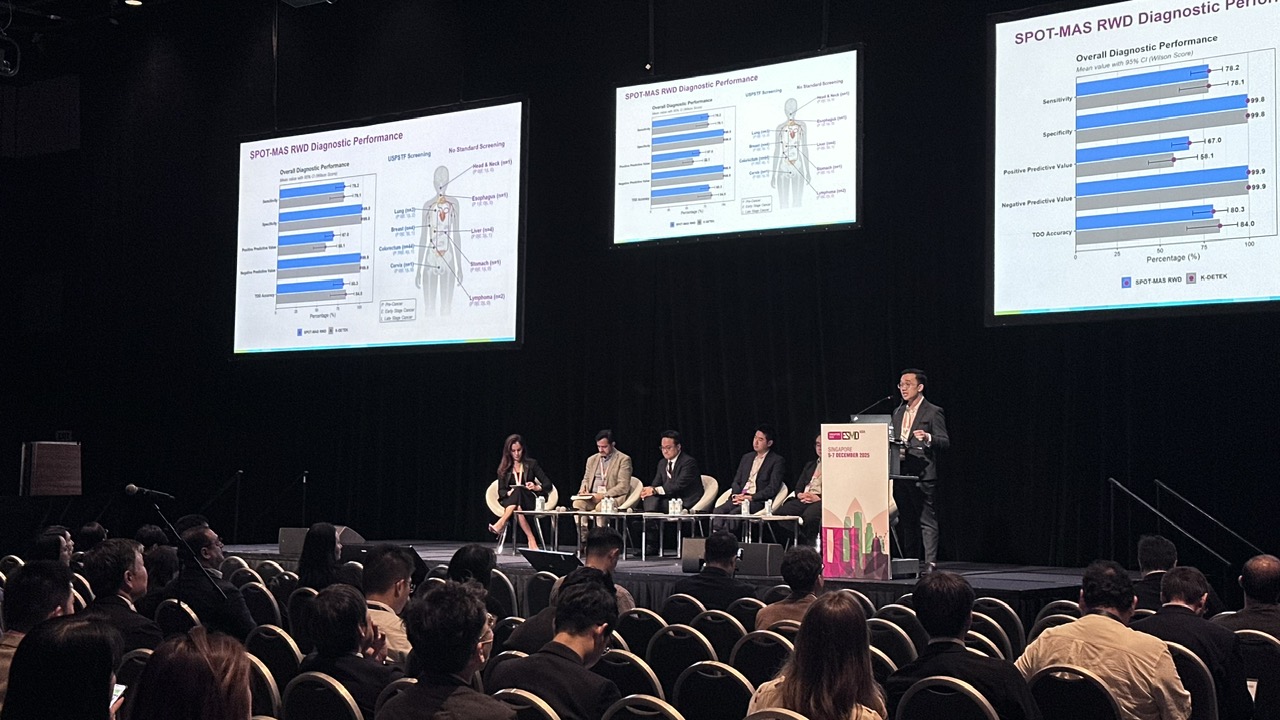
Dr. Nguyen concluded with a strategic roadmap: refining AI for complex biological backgrounds, expanding screening to high-risk genetic cohorts, and validating long-term survival and health-economic benefits.
Audience discussion probed biological limits – what if very early tumors shed minimal ctDNA? Dr. Tran acknowledged: “Not all tumors shed DNA,” noting that aggressiveness correlates with shedding-level, and detection may prioritize clinically consequential cancers. Prof. Aya El Helali added a forward-looking perspective: “To push stage I sensitivity in multi-cancer early detection, we must integrate multi layered AI such as methylation, fragmentomics—it is essential when tumors shed little ctDNA.”
ctDNA MRD in Action: Lung Cancer Focus
Speaker: Asst. Prof. Aaron C. Tan – ctDNA MRD in action: Current applications to emerging horizons

Asst. Prof. Tan situated MRD within the rapidly evolving early‑stage NSCLC landscape—now populated by adjuvant targeted therapies, immunotherapy, neoadjuvant, and perioperative regimens. “In early-stage cancer, cancers may still recur in some patients despite adjuvant treatment, while some patients may already be cured and do not need additional adjuvant therapy. MRD testing has the potential to transform how we manage these patients by allowing clinicians to escalate or de-escalate treatment more appropriately.” he said, while cautioning that assay sensitivity remains challenging in lung cancer and harmonization is essential.
He contrasted tumor‑informed bespoke assays (leveraging WES/WGS to personalize detection) with tumor‑agnostic strategies (methylation/fragmentomics), advocating for multi‑omics integration to improve sensitivity and PPV. In a de‑identified illustrative scenario, a patient with multiple synchronous, early-stage primaries underwent NGS revealing distinct somatic drivers; ctDNA‑MRD was negative, supporting a surveillance‑only approach over adjuvant therapy through shared decision‑making.
Advanced AI‑Driven Biomarkers: Integration & Standards

Prof. Herbert Ho Fung Loong defined AI‑driven biomarkers as indicators of cancer presence, behavior, or treatment response “discovered, quantified, or interpreted using machine learning or deep learning.” He outlined how these biomarkers are already shaping, or have the potential to shape, decisions across multiple stages of cancer care:
- Screening and early detection: AI-assisted CT imaging is refining lung cancer screening by scoring malignancy risk, while fragmentomics is enabling multi-cancer early detection (MCED) from a simple blood draw.
- Treatment selection (predictive biomarkers): Radiomic or pathomic signatures of inflamed vs. immune-excluded tumour microenvironment to predict immunotherapy benefit.
- Prognosis/risk stratification: AI-enhanced ctDNA analysis improves minimal residual disease (MRD) detection, guiding adjuvant therapy decisions.
- Treatment response monitoring: Tools like AI-RECIST detect subtle imaging changes before tumors shrink, and predictive algorithms are emerging to forecast toxicities such as neutropenia or cardiotoxicity.
To illustrate practical application, Prof. Loong showcased K-4CARE, Gene Solutions’ AI-powered multi-omics platform. This triad approach combines: DNA profiling (504 genes), transcriptomic profiling (19,435 genes), and longitudinal ctDNA-MRD monitoring to support comprehensive precision decisions, even in challenging scenarios such as cancers of unknown primary (CUP). The platform achieved 87.7% tissue-of-origin prediction accuracy for CUP and improved fusion detection by 20%, underscoring its clinical utility. (2)
He concluded by introducing the ESMO Basic Requirements for AI-Based Biomarkers in Oncology (EBAI), a framework that sets the bar for robustness: clarity of ground truth, proven performance, generalizability, and fairness – ensuring AI-driven tools move beyond theoretical promise to deliver real-world clinical impact. Prof. Loong closed with a vision that resonated across the room: “AI-driven biomarkers won’t replace molecular testing; they will amplify it – especially when tissue is limited.”
Panel Discussion Takeaways: From Evidence to Practice in Asia
Prof. Aya El Helali shared a candid perspective: advanced/metastatic settings already benefit from ctDNA for early progression, IO response assessment, and treatment adaptation. In earlier‑stage disease, tumor context matters; colorectal stage II shows stronger support than stage III for de‑escalation strategies, while lung needs more prospective escalation/de‑escalation trials.
Prof. Pavlakis and Prof. Tan reiterated the importance of shared decision‑making amid evolving evidence: action on MRD must consider assay limits, disease biology, and patient preferences. Prof. Loong emphasized real‑world data feedback loops to refine assays and regional collaboration to accelerate adoption.
Looking Forward
In an era where cancer biology is multi‑layered and dynamic, the convergence of AI, liquid biopsy, and multi‑omics offers clinicians a sharper lens on risk, response, and resistance. As the symposium made clear, the path forward in Asia will be defined by rigorous validation, thoughtful clinical integration, and collaboration—turning technological promise into measurable patient benefit.
References:
- Van-Anh Nguyen Hoang et al. JCO Oncology Adv (2025)
- Bao-Nam Tran Huynh et al. Cancer Medicine (2025)
