Gene Solutions Indonesia and PDPI Banten Hold a Workshop on Lung Cancer Treatment Innovation
On September 22nd, 2024, Gene Solutions Indonesia, in collaboration with the Indonesian Society of Respirology (PDPI) Banten branch, held a hybrid workshop titled “The Future of Cancer Management: Implementation of Advanced Technology on Cancer Care.” Focused on lung cancer – Indonesia’s second most common cancer – the event was the culmination of a roadshow aimed at educating thoracic oncologist about the latest technological advances in cancer treatment.
Dr. Sita L. Andarini, Ph.D., Sp. P(K) presenting

Dr. M. Alfin Hanif, SpP (K) Onk presenting
Keynote speaker Associate Prof. dr. Sita Laksmi Andarini, Ph.D, SpP(K), from National Respiratory Center, shared insights into the latest innovations in lung cancer detection and treatment. Meanwhile, dr. Alfin Hanif, Sp.P(K)Onk from National Cancer Center, followed with his experiences in managing lung cancer cases, highlighting the application of cutting-edge technology in cancer care.

Country Director introduced Gene Solutions Indonesia
Gene Solutions Indonesia also presented two innovative technologies. The first is K-TRACK, a cost-effective and personalized ctDNA assay that can be applied to all stages of lung cancer patients. For early-stage patients, with its high sensitivity and specificity (87.5% and 95%, respectively), K-TRACK can monitor post-treatment progress and predict cancer relapse, with a lead time of up to 14.4 months. For advanced-stage patients, ctDNA is especially useful for those receiving targeted therapies against driver oncogenes or immunotherapy.
Online participant overview

Q&A Session

Q&A Session
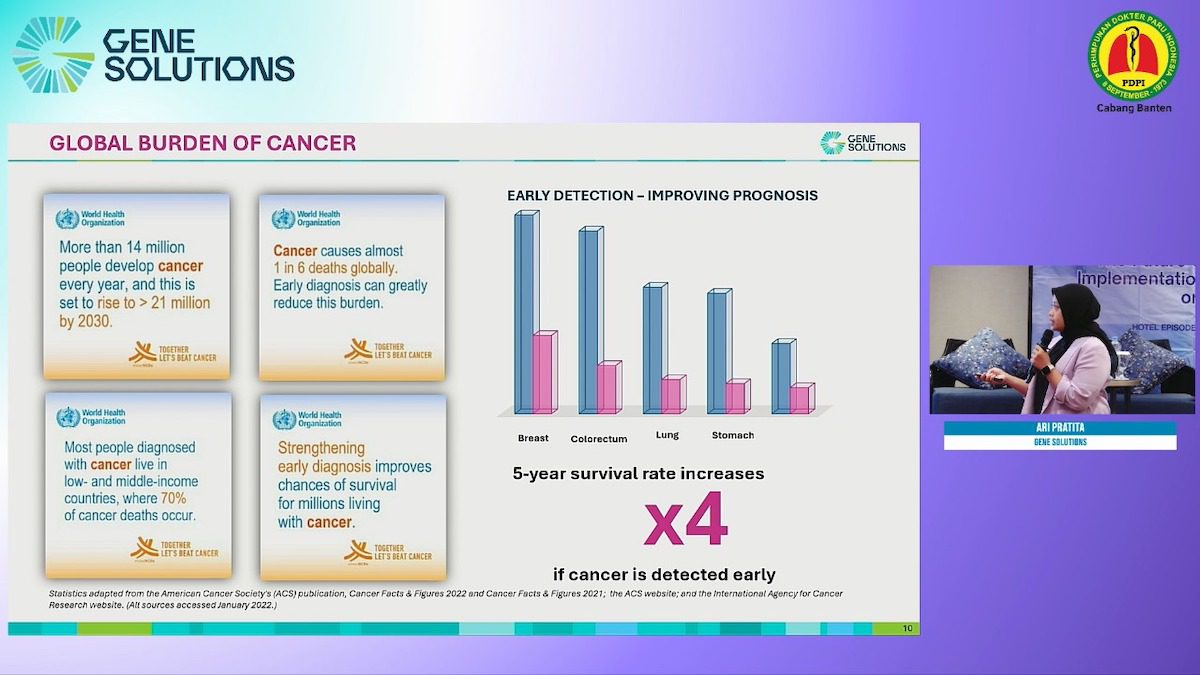
The second innovative product is SPOT-MAS, a non-invasive test that incorporates Next-Generation Sequencing (NGS) and AI-analytics technology to detect ctDNA signals from lung cancer at an early stage with high sensitivity (83.7%). It also detects ctDNA signals from other common cancer types, providing numerous treatment options at early stages and positive prognostic outcomes. Both innovative technologies garnered positive feedback from in-person and online attendees, especially regarding the real-world implementation in lung cancer care.

Closing statement from DR. dr. Tri Agus Yuarsa, Sp.P(K)Onk, MKes, MARS as the Chair of PDPI Banten branch
The workshop concluded with remarks from Dr. dr. Tri Agus Yuarsa, Sp.P(K)Onk, MKes, MARS as the Chair of PDPI Banten branch, stressing the value of complementary screening methods in lung cancer. This additional screening provides essential perspectives, helping doctors offer more comprehensive and optimal cancer care for patients.
About Gene Solutions Indonesia:
Established in 2022, PT Gene Solutions Indonesia has been diligently advancing genetic testing and providing essential support to healthcare practitioners. We recognize the significance of Next-Generation Sequencing (NGS) in prenatal care and molecular diagnostics. Through our valued partnership with KALGen INNOLAB, we strive to make Non-Invasive Prenatal Testing (NIPT) and oncology diagnostics accessible to the Indonesian population.
Follow Gene Solutions Indonesia on Instagram to get latest update. https://www.instagram.com/genesolutions.id/
