Gene Solutions’ Presentations at ASCOMOS 2024
Fueled by a mission to advance cancer care, Gene Solutions recently joined the 35th Annual Scientific Congress of the Malaysian Oncological Society (ASCOMOS 2024) as a Gold Sponsor. Held from October 11-13 at the Setia SPICE Convention Centre in Penang, the gathering brought together over 1,095 healthcare professionals who shared a collective passion for transforming oncology. As a proud Gold Sponsor, we not only showcased our commitment to innovation but also engaged in vital discussions about the future of cancer treatment.


Highlights in Multi-Cancer Early Detection and Personalized Cancer Tracking
With a focus on Multi-Cancer Early Detection (MCED) and Minimal Residual Disease (MRD), our R&D and Medical teams introduced latest updates from K-TRACKTM MRD and SPOT-MASTM MCED test, showcasing their real-world applications and clinical impact.
1. Gene Solutions Symposium: The Dynamic Role of ctDNA in Cancer Monitoring | Speaker: Dr. Duy-Sinh Nguyen PhD, MD | K-TRACKTM
During the symposium titled “The Dynamic ctDNA: Minimal Residual Disease & Beyond – Monitoring & Clinical Applications,” Dr. Duy-Sinh Nguyen, PhD, MD, Medical Director at Gene Solutions, shared valuable insights into the growing clinical importance of circulating tumor DNA (ctDNA) for personalized cancer care. He highlighted the latest results from clinical trials on the predictive value of ctDNA in escalate/de-escalate adjuvant therapies and its solid prognostic benefits in recurrence monitoring compared to conventional methods like CEA or imaging.
“During a 3-year follow-up of post-operative colorectal cancer in our recent publication on ESMO Real World Data & Digital Oncology, ctDNA showed a 100% positive predictive value (PPV) in detecting recurrence, with a median lead time of 8 months before clinical diagnosis. In contrast, the PPV of Carcinoembryonic Antigen (CEA) is 50%. Furthermore, ctDNA was more sensitive and specific than CEA in detecting recurrence,” said Dr. Duy-Sinh Nguyen.
The symposium generated significant interest from oncologists in ctDNA’s potential for guiding treatment adjustments based on individual risk profiles.

2. Oral presentation + Physical Poster: Personalized Mutation Tracking in Circulating-tumor DNA Predicts Recurrence in Patients with High-risk Early Breast Cancer | Abstract #A-0147 | Presenter: Van-Anh Nguyen Hoang, MSc | K-TRACKTM
Gene Solutions presented compelling data demonstrating that patients with circulating tumor DNA (ctDNA) positivity at any point after surgery had a significantly higher risk of cancer recurrence. With a median lead time of 9.7 months before clinical diagnosis (up to 13.2 months), K-TRACK™ promises to revolutionize breast cancer recurrence monitoring.

3. Physical poster: A Streamlined Personalized Assay To Detect Molecular Residual Disease In Solid Tumors | Abstracts #A-0148 | Presenter: Bich-Ngoc Nguyen Thi, BSc | K-TRACKTM
This research emphasizes K-TRACK™’s utility in monitoring minimal residual disease (MRD) and actionable mutation profiling, paving the way for affordable precision oncology.
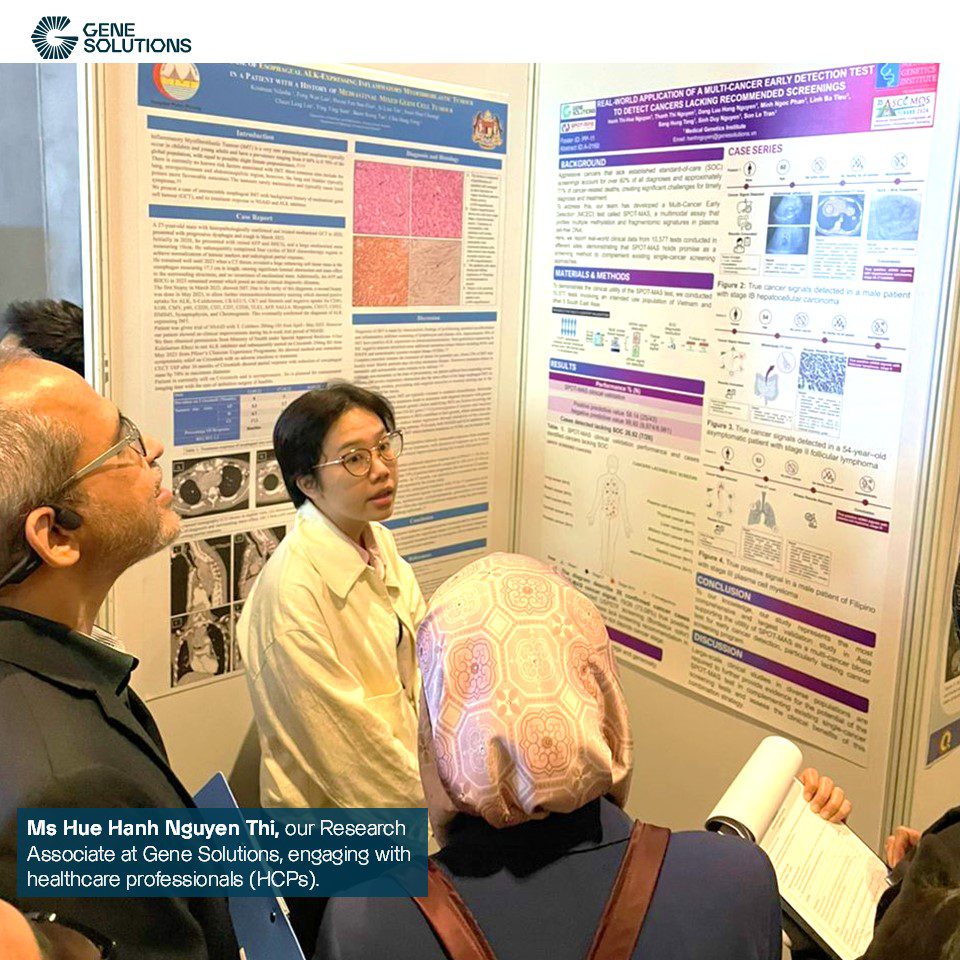
4. Poster presentation + Physical Poster: Real-world Application of A Multi-Cancer Early Detection Test to Detect Cancers Lacking Recommended Screening | Abstract #A-0160 | Hue-Hanh Nguyen Thi, BSc | SPOT-MASTM
Data from over 10,500 tests of SPOT-MAS™, a multimodal assay that profiles methylation and fragmentomic signatures in plasma cell-free DNA for Multi-cancer early detection, demonstrated its effectiveness in complementing existing single-cancer screening methods. With a Positive Predictive Value (PPV) of 58.14% and a Negative Predictive Value (NPV) of 99.92%, SPOT-MAS™ holds great potential for detecting cancers that lack recommended screening options.
Summary:
Gene Solutions’ Malaysia participation at ASCOMOS 2024 showcased the valuable role of emerging technologies in oncology. Each presentation reflected our commitment to pushing forward the frontiers of cancer care, with a vision of broadening precision oncology’s impact on patient outcomes across Malaysia and the wider region.

About Gene Solutions Malaysia
Gene Solutions established its operations and services in Malaysia in 2023 and is committed to provide the local community access to next-generation genetic tests that support reproductive health and clinical oncology. In the field of clinical oncology, personalized minimal residual disease (MRD) detection, and multi-cancer early detection (MCED) tests using advanced ctDNA technology are among some of the products available. Partnering with local laboratory partners Premier Integrated Lab and Innoquest Malaysia, Gene Solutions Malaysia is committed to enhancing healthcare with their innovative genetic solutions.
