Lead R&D Scientist to Present Breakthrough Multi-Cancer Early Detection Research Findings at Prestigious ASCO Annual Meeting 2024 in Chicago
As a significant development for the MCED community, Dr. Le Son Tran, the lead R&D scientist from Medical Genetics Institute Ho Chi Minh City and Gene Solutions is set to present breakthrough findings at the upcoming prestigious oncology conference this weekend.
The landscape of MCED tests based on detecting methylation changes in cfDNA has evolved but current solutions still exhibit low sensitivity for detecting certain cancers (e.g., breast cancer) and early-stage tumors owing to low amount and high heterogeneity of ctDNA. To improve the detection sensitivity of ctDNA, MCED screening methods tend to use high-depth sequencing, making it economically impractical for population-wide screening. To address these limitations, the team at Medical Genetics Institute Ho Chi Minh City and Gene Solutions developed a novel approach – Screening for the Presence Of Tumor by Methylation And Size (SPOT-MAS), which offers a comprehensive analysis through the integration of methylation, fragment length profile, DNA copy number aberration and end motif in a single library reaction. It was designed to simultaneously detect five common types of cancer, including liver, breast, colorectal, gastric, and lung cancer, and predict the tissue origin of cancer signal1. The performance and findings from a multi-centre prospective study support the utility of SPOT-MAS as a multi-cancer blood test for early cancer detection, particularly in low- and middle-income countries lacking nationwide cancer screening programs2.
Innovative Research: The scientist will discuss innovative approaches to ctDNA analysis using SPOT-MAS for Multi-Cancer Early Detection of asymptomatic individuals, potentially revolutionizing cancer preventive care.
Rigorous Analytical and Clinical Validation: The team undertook the K-DETEK multi-centre prospective study to validate the diagnostic performance of SPOT-MAS, recruiting 10,027 asymptomatic individuals aged 40 years or older.
Future of Oncology: The strong performance of SPOT-MAS in the K-DETEK study underpins the potential to shape the future direction of oncology research and treatment.
The research team welcomes collaborative opportunities and anticipates our participation at ASCO Annual Meeting will provide a platform for collaboration among top oncology professionals.
The conference will run from May 31 to June 4, at McCormick Place, Chicago USA and is also available online. It is an opportunity not to be missed for anyone interested in the forefront of cancer research. For more information and to register for the event, please visit https://conferences.asco.org/am/attend.
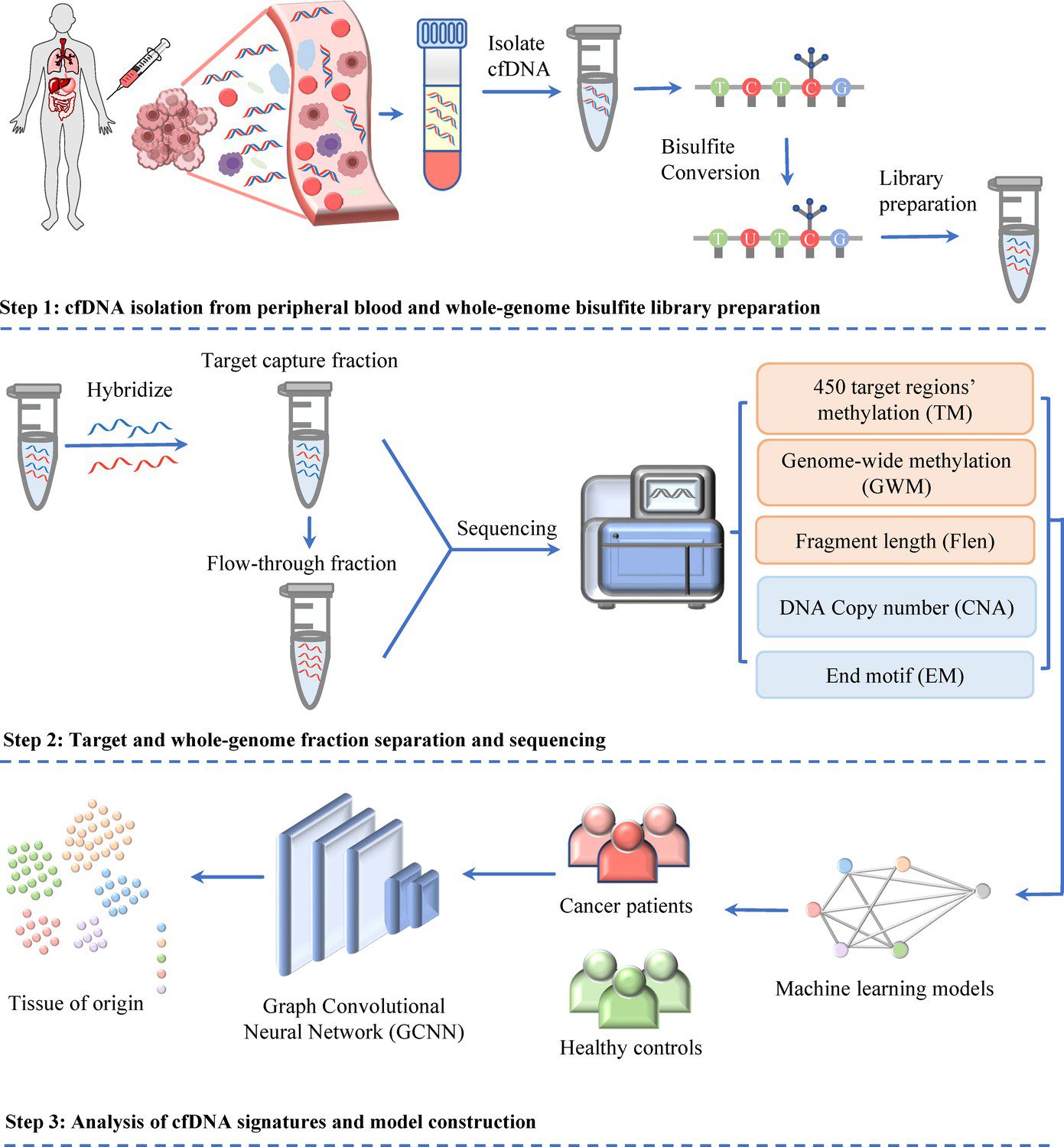
Workflow of SPOT-MAS (screening for the presence of tumor by methylation and size) assay for multi-cancer detection and localization1.
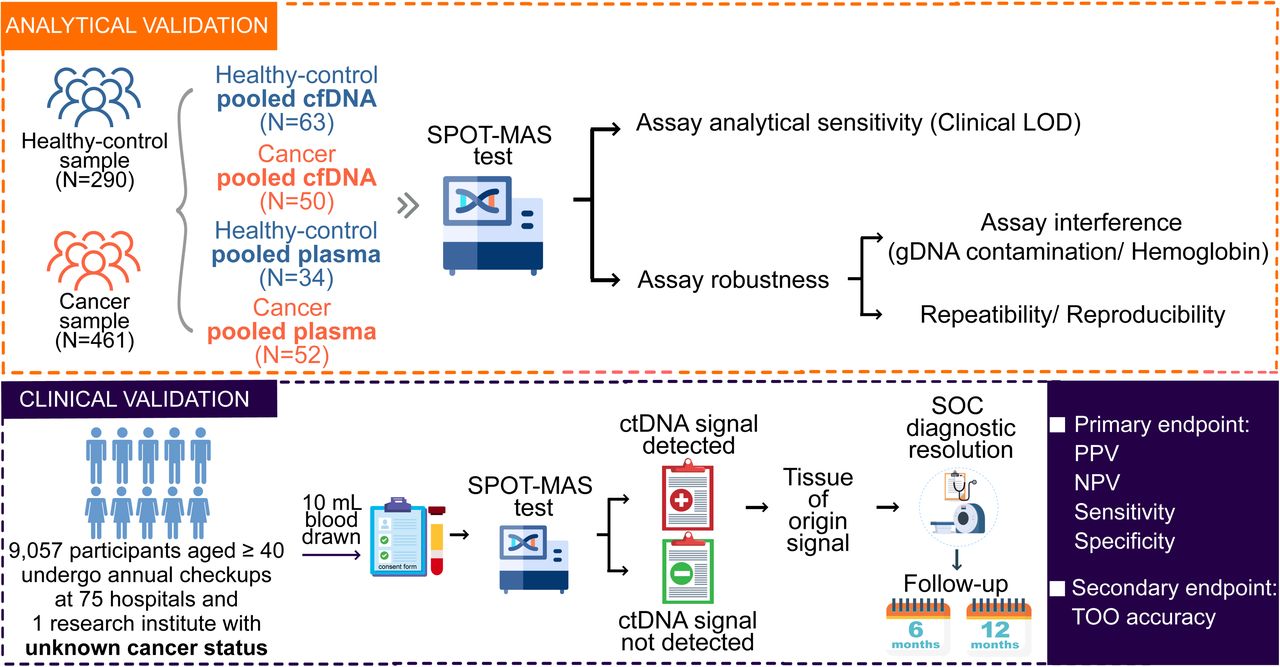 Overview of analytical and clinical validation2
Overview of analytical and clinical validation2
- Van Thien Chi Nguyen, et al (2023) Multimodal analysis of methylomics and fragmentomics in plasma cell-free DNA for multi-cancer early detection and localization eLife 12:RP89083 https://doi.org/10.7554/eLife.89083.3
- Luu Hong Dang Nguyen et al, Analytical and clinical validation of a circulating tumor DNA-based assay for multi-cancer early detection. medRxiv 12.22.23300420; doi: https://doi.org/10.1101/2023.12.22.23300420
